UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
(State
or other jurisdiction |
(Commission |
(IRS Employer |
| (Address of principal executive offices) | (Zip Code) |
(Registrant’s telephone number, including
area code): (
Not Applicable
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Name Of Each Exchange Trading Symbol(s) |
On Which Registered | ||
| Global Market | ||||
| Global Market* |
* Not for trading, but only in connection with the registration of the American Depositary Shares
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 7.01 Regulation FD Disclosure.
On December 8, 2025, Structure Therapeutics Inc. (the Company) issued a press release announcing data from its ACCESS clinical program of aleniglipron, the company’s once-daily oral small molecule glucagon-like-peptide-1 (GLP-1) receptor agonist (GLP1-RA) for the treatment of obesity. A copy of the press release is furnished as Exhibit 99.1 to this Current Report on Form 8-K.
On December 8, 2025, the Company made available on its website an investor presentation to be shared with investors and others from time to time. A copy of this presentation is being furnished as Exhibit 99.2 to this Current Report on Form 8-K.
The information set forth in this Item 7.01 and in the press release and investor presentation attached hereto as Exhibits 99.1 and 99.2, respectively, is deemed to be “furnished” and shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the Exchange Act), or otherwise subject to the liabilities of that Section. The information set forth in this Item 7.01, including Exhibits 99.1 and 99.2, shall not be deemed incorporated by reference into any filing under the Exchange Act or the Securities Act of 1933, as amended, except to the extent that the Company specifically incorporates it by reference.
Item 8.01 Other Events.
Summary of Data from the ACCESS Clinical Program
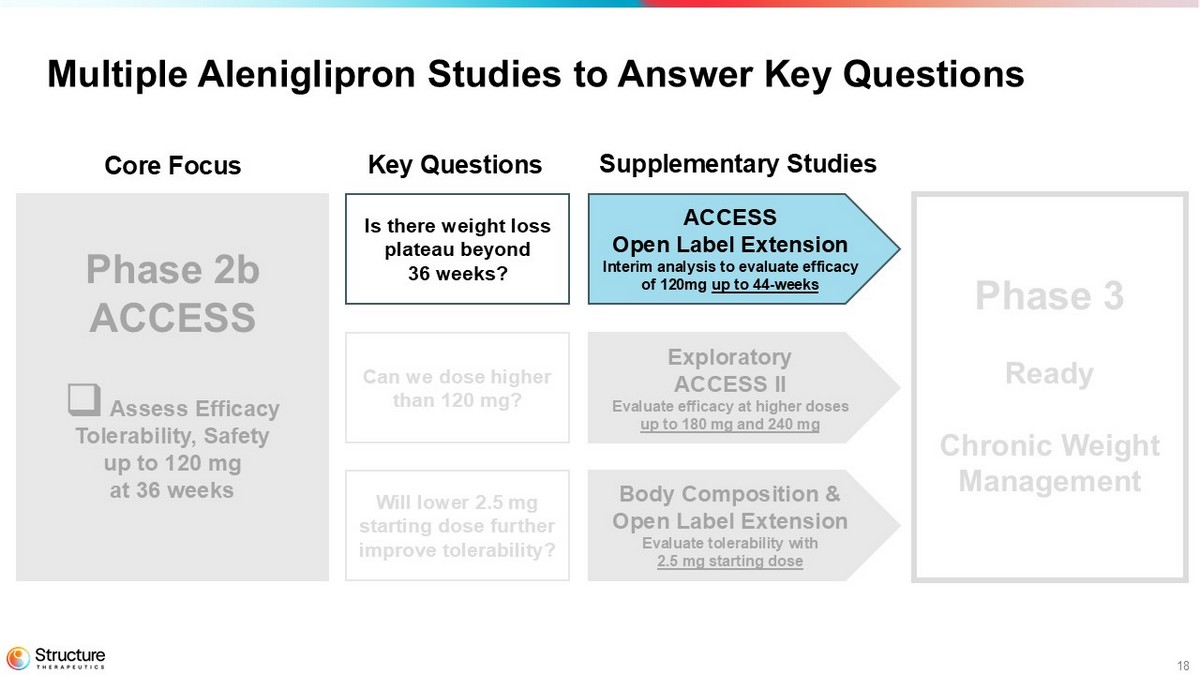
The Company’s ACCESS clinical program includes 36-week data from the core Phase 2b ACCESS study and exploratory ACCESS II study, and interim data from Phase 2 body composition study and Phase 2b ACCESS open label extension (OLE) study, as follows:
| • | The Phase 2b ACCESS study demonstrated a placebo-adjusted mean weight loss of 11.3% with 120 mg dose at 36 weeks. |
| • | The exploratory ACCESS II dose exploration study demonstrated a placebo-adjusted mean weight loss of 15.3% at 240 mg at 36 weeks. |
| • | No adverse event-related treatment discontinuations were observed when starting at the lower 2.5 mg dose in ACCESS Open Label Extension and Body Composition Study |
| • | The Company believes that the data from the ACCESS clinical program supports and informs the advancement to Phase 3 clinical development program in mid-2026. |
The description of each study and the data observed is set forth below.
Phase 2b ACCESS study - Evaluating target doses of up to 120 mg
The core 36-week Phase 2b ACCESS study was a randomized, double-blind, placebo-controlled, Phase 2b dose-range finding clinical study that enrolled 230 adult participants living with obesity (body mass index (BMI) ≥ 30 kg/m2), or overweight (BMI ≥ 27 kg/m2) with at least one weight-related comorbidity. Participants were enrolled in 36 participating sites across the United States. Participant ages ranged from 49 to 52 years and participants were predominantly female (53% to 55%) with a baseline BMI of 39. HbA1c and blood pressure, according to the eligibility criteria, were within normal limits. All participants were randomized 3:1 (active:placebo) and started at 5 mg of aleniglipron (or placebo) with a 4-week titration schedule, reaching target doses of 45 mg, 90 mg or 120 mg once-daily.
Each of the three doses in the ACCESS study achieved statistical significance on the primary endpoint and all key secondary endpoints. Primary efficacy estimand1 results at 36 weeks are as follows:
Aleniglipron 45 mg |
Aleniglipron 90 mg |
Aleniglipron 120 mg |
Placebo | |
| Mean percent change in body weight at 36 weeks compared to baseline | -9.0 | -10.7 | -12.1 | -0.8 |
| Placebo-adjusted mean percent change in body weight at 36 weeks compared to baseline | -8.2 | -9.8 | -11.3 | - |
| P-value | p<0.0001 | p<0.0001 | p<0.0001 | - |
At week 36, key secondary endpoints in the study show that 86% of participants in the aleniglipron 120 mg dose cohort achieved at least 5% weight loss and 70% achieved at least 10% weight loss. In addition, aleniglipron demonstrated clinically meaningful improvements in systolic blood pressure (-6.4 to -7.5 mmHg) and HbA1c (-0.28% to -0.37%).
Aleniglipron demonstrated a tolerability profile consistent with the GLP-1 receptor agonist class following repeated, once-daily dosing of up to 120 mg in the Phase 2b ACCESS study. As expected for the GLP1-RA drug class, the most common AEs were gastrointestinal (GI)-related and the two most common AEs in the titration phase were nausea and vomiting. AEs were generally observed early in treatment. In the Phase 2b ACCESS study, the AE-related treatment discontinuation rate ranged from 7.7% - 13.3% between all doses, with a mean 10.4% across all active arms in the study.
Exploratory ACCESS II Study - Evaluating higher doses up to 240 mg
ACCESS II is a randomized, double-blind, placebo-controlled, clinical study of aleniglipron that enrolled 85 adult participants living with obesity, or overweight with at least one weight-related comorbidity. Participants were enrolled in ten sites across the United States. The study was designed to evaluate two higher doses of aleniglipron. Participants started at 5 mg of aleniglipron (or placebo) and followed a 4-week titration schedule up to target doses of 120 mg, 180 mg and 240 mg. Twelve participants were part of the sentinel group which aimed to provide preliminary data at the 180 mg/day dose to the independent data monitoring committee before moving to higher doses in the main part of the study, which included 73 participants, 61 allocated to aleniglipron and 12 to placebo. At week 28, the remaining participants at 120 mg of aleniglipron were rerandomized to stay on 120 mg, titrate to 180 mg, or titrate to 180 mg to ultimately reach 240 mg. The 44-week study remains ongoing with data from a prespecified 36-week analysis currently available.
At 36 weeks, each of the three dose cohorts in the ACCESS II study met statistical significance compared to placebo. Primary efficacy estimand results at 36 weeks are as follows:
Aleniglipron 120 mg |
Aleniglipron 180 mg |
Aleniglipron 240 mg |
Placebo | |
| Mean percent change in body weight at 36 weeks compared to baseline | -13.1 | -13.3 | -14.2 | +1.0 |
| Placebo-adjusted mean percent change in body weight at 36 weeks compared to baseline | -14.1 | -14.4 | -15.3 | - |
| P-value | p<0.0001 | p<0.0001 | p<0.0001 | - |
Aleniglipron demonstrated a tolerability profile consistent with the GLP1-RA class following repeated, once-daily dosing of up to 240 mg. As expected for the GLP1-RA drug class, the most common AEs were gastrointestinal (GI)-related and the two most common AEs in the titration phase were nausea and vomiting. AEs were generally observed early in treatment.
1 The primary efficacy estimand represents efficacy had all randomized participants remained on study treatment (with possible dose interruptions and/or dose modifications) for 36 weeks without initiating rescue weight management treatments or surgeries.
Body Composition Study - Evaluating lower 2.5 mg starting dose
The Company is currently conducting a randomized, placebo-controlled body composition study that enrolled 71 adult participants to assess the effect of aleniglipron (up to 120 mg) on body fat loss over a 40-week evaluation period. Participants with similar eligibility criteria to the previous studies were enrolled in 11 sites in the United States. 59 participants were randomized to the aleniglipron treatment arm, started at a 2.5 mg dose, and titrated up monthly to a target dose of 120 mg. Data from a pre-specified interim analysis after a median follow-up time of approximately 10 weeks showed that starting at a lower dose of 2.5 mg for the first four weeks meaningfully improved tolerability compared to what was observed at a starting titration dose of 5 mg in the ACCESS and ACCESS II studies, with no AE-related treatment discontinuations observed at the initial 2.5 mg dose or the subsequent 5 mg dose.
ACCESS OLE Study - Following randomized 36-week period, evaluating lower 2.5mg starting dose
Following the 36-week randomized controlled portion of the Phase 2b ACCESS study, the majority of eligible ACCESS participants enrolled in ACCESS OLE. An initial analysis from the ongoing OLE demonstrates continuing weight loss in all dose cohorts out to 44 weeks, showing no evidence of weight loss plateau and proportional pharmacokinetic exposure up to 240 mg. Participants in the Phase 2b ACCESS study are provided an option to roll over into an OLE and receive aleniglipron for an additional 36 weeks.
In the ACCESS OLE study, participants who received placebo in the initial double-blind portion transitioned to aleniglipron at a starting dose of 2.5 mg and titrate monthly to a target dose of 120 mg. Initial data from this group of participants after eight weeks of treatment are consistent with the findings from the body composition study, showing that starting at a 2.5 mg titration dose meaningfully improved tolerability compared to what was observed in the starting 5 mg titration dose in ACCESS and ACCESS II studies, with no AE-related treatment discontinuations at the initial 2.5 mg or the subsequent 5 mg dose.
Aleniglipron Safety
Aleniglipron demonstrated a compelling safety profile across all studies. Importantly, there were no cases of drug-induced liver injury, no persistent liver enzyme elevations, and no QTc prolongation across all aleniglipron studies.
Phase 3 Preparation
Data from ACCESS, ACCESS II, body composition, and the ACCESS OLE studies provide a strong foundation to advance aleniglipron into Phase 3 clinical development and the Company expects to report topline results for these studies in the first half of 2026. The Company plans to request a Type B End-of-Phase 2 meeting with the U.S. Food and Drug Administration (FDA) in the first half of 2026 to finalize the Phase 3 design, which is currently designed with a starting titration dose of 2.5 mg with the intent to evaluate multiple doses up to 240 mg. The Company anticipates initiating the Phase 3 program by mid-2026.
In the second half of 2026, the Company expects to report topline results from its maintenance switching study evaluating the transition from an approved injectable GLP1-RA injectable to aleniglipron for weight loss maintenance (SWITCH study), and its Phase 2 randomized placebo controlled study assessing aleniglipron at doses of up to 240mg in patients living with obesity or overweight and type 2 diabetes mellitus (T2DM study). Additionally, the Company expects to receive Phase 1 study results for ACCG-2671, its dual amylin calcitonin receptor agonist (DACRA) and to initiate a Phase 1 study for its second DACRA development candidate, ACCG-3535, in the second half of 2026.
Forward-Looking Statements
This Current Report on Form 8-K contains “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical fact are statements that could be deemed forward-looking statements, including, without limitation, statements concerning: the Company’s future plans and prospects; any expectations regarding the potential benefits, tolerability and safety profile, accessibility, scalability, combinability, capability, efficacy, convenience, expected effects and future application of aleniglipron; the belief that data to date from the Phase 2b ACCESS, exploratory ACCESS II, body composition, and the Phase 2b ACCESS OLE studies support and inform advancement of the Phase 3 clinical development of aleniglipron; the expected timing for the meeting with the FDA to finalize the Phase 3 trial design and the Phase 3 program initiation of aleniglipron; the expected timing of topline data readouts from the exploratory ACCESS II, body composition, the Phase 2b ACCESS OLE, SWITCH and T2DM studies; the planned initiation of the ACCG-3535 Phase 1 study and the timing thereof; the expected timing of study results from the Phase 1 ACCG-2671 study; and any presumption that topline, interim or preliminary data will be representative of final data or data in later clinical trials. In addition, when or if used in this Current Report on Form 8-K, the words and phrases “anticipate,” “believe,” “expect,” “may,” “plan,” “potential,” “to be,” “will,” and similar expressions and their variants, as they relate to the Company may identify forward-looking statements. Forward-looking statements are neither historical facts nor assurances of future performance. Although the Company believes the expectations reflected in such forward-looking statements are reasonable, the Company can give no assurance that such expectations will prove to be correct. Readers are cautioned that actual results, levels of activity, safety, performance or events and circumstances could differ materially from those expressed or implied in the Company’s forward-looking statements due to a variety of risks and uncertainties, which include, without limitation: risks and uncertainties related to topline results that the Company reports are based on preliminary analysis of key efficacy and safety data, and such data may change following a more comprehensive review of the data related to the clinical trial and such topline data may not accurately reflect the complete results of a clinical trial, the preliminary nature of the results due to the length of the study and sample size and the results from earlier clinical studies not necessarily being predictive of future results; potential delays in the commencement, enrollment and completion of the Company’s planned clinical studies; the Company’s ability to advance aleniglipron, ACCG-2671, ACCG-3535, ANPA-0073, LTSE-2578, and its other therapeutic candidates, obtain regulatory approval of, and ultimately commercialize the Company’s therapeutic candidates; competitive products or approaches limiting the commercial value of the Company’s product candidates; the timing and results of preclinical and clinical studies; the Company’s ability to fund development activities and achieve development goals; the Company's reliance on third parties, including clinical research organizations, manufacturers, suppliers and collaborators, over which it may not always have full control; general geopolitical and macroeconomic conditions, including as a result of tariffs and various global conflicts; the Company’s ability to protect its intellectual property; and other risks and uncertainties described in the Company’s filings with the U.S. Securities and Exchange Commission (SEC), including the Company’s latest Quarterly Report on Form 10-Q and future reports the Company may file with the SEC from time to time. All forward-looking statements contained in this Current Report on Form 8-K speak only as of the date on which they were made and are based on management’s assumptions and estimates as of such date. The Company undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by law.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits.
| Exhibit No. |
Description | |
| 99.1 | Press Release, dated December 8, 2025. | |
| 99.2 | Investor Presentation, dated December 8, 2025. | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Structure Therapeutics Inc. | ||
| Date: December 8, 2025 | By: | /s/ Raymond Stevens |
| Raymond Stevens, Ph.D. | ||
| Chief Executive Officer | ||
Exhibit 99.1

Structure Therapeutics Reports Positive Topline Data from ACCESS Program for its Once-Daily Oral Small Molecule GLP-1 Receptor Agonist, Aleniglipron
Placebo-adjusted mean weight loss of 11.3% (27.3 lbs) with 120 mg dose in the 36-week Phase 2b ACCESS study with a 10.4% adverse event-related treatment discontinuation
Placebo-adjusted mean weight loss up to 15.3% (35.5 lbs) observed with 240 mg dose in the exploratory ACCESS II study at 36 weeks
No adverse event-related treatment discontinuations observed when starting at lower 2.5 mg dose in ACCESS Open Label Extension and Body Composition Study
Data comprehensively support and inform advancement to Phase 3 clinical development program in mid-2026
Company to host conference call today at 8:30 a.m. Eastern Time
SAN FRANCISCO, December 8, 2025– Structure Therapeutics Inc. (NASDAQ: GPCR), a clinical-stage global biopharmaceutical company developing novel oral small molecule therapeutics for metabolic diseases, with a focus on obesity, today announced positive topline data from the ACCESS clinical program of aleniglipron for the treatment of people living with obesity and/or overweight with at least one weight related co-morbidity. This includes 36-week data from the core Phase 2b ACCESS study and the ongoing exploratory ACCESS II study, and interim data from the ongoing Body Composition study and the ACCESS open label extension (OLE) study. Aleniglipron is an investigational orally-available, once-daily, nonpeptide small molecule agonist of the glucagon-like-peptide-1 (GLP-1) receptor designed to address patient needs and accessibility.
In the core Phase 2b ACCESS study, aleniglipron achieved a clinically meaningful and statistically significant placebo-adjusted mean weight loss of 11.3% (27.3 lbs, p<0.0001) at the 120 mg dose at 36 weeks and across all active arms had a 10.4% adverse event (AE)-related treatment discontinuation rate. In the exploratory ACCESS II study, aleniglipron achieved a placebo-adjusted mean weight loss up to 15.3% (35.5 lbs, p<0.0001) with 240 mg dose at 36 weeks. Aleniglipron demonstrated a tolerability profile consistent with the GLP-1 receptor agonist class, and a compelling off-target safety profile. Together, these positive findings support the advancement of aleniglipron into Phase 3 clinical development.
“The topline results presented today show that aleniglipron is differentiated and delivered clinically meaningful, competitive and dose-dependent weight loss with a safety profile appropriate for chronic use in a disease that impacts millions of people,” said Raymond Stevens, Ph.D., CEO of Structure Therapeutics. “For the higher doses, the observed weight loss data at 36 weeks with no weight loss plateau is potentially best-in-class for oral small molecule GLP1s. Most importantly, these findings provide comprehensive information to move into Phase 3 development and reinforce aleniglipron’s potential to become a backbone oral small molecule therapy for obesity—one that is accessible, scalable, and combinable.”
“The weight-lowering data from these ACCESS studies, without any evidence of a plateau by Week 36, are very encouraging—particularly weight loss of up to 15.3% in ACCESS II that hopefully will be confirmed in larger, longer-term studies,” said Julio Rosenstock, MD, Chair of the ACCESS program Steering Committee and Senior Scientific Advisor of Velocity Clinical Research and Clinical Professor of Medicine, Univ. of Texas, Southwestern Medical Center. “As once-daily oral, non-peptide, small molecule GLP-1 RAs such as aleniglipron become available, they have the potential to transform obesity treatment and broaden access, which could have a profound impact on patients globally.”
“Obesity is a complex, chronic disease, and far too many individuals still face barriers to accessing effective, long-term treatment options,” said Joe Nadglowski, Obesity Action Coalition President and CEO. “A once-daily oral therapy like aleniglipron has the potential to expand treatment options for people living with obesity. The results from the ACCESS programs represent a promising advance in the therapeutic landscape and bring us closer to a future where people living with obesity have multiple, accessible options to address their needs.”
Phase 2b ACCESS study - Evaluating target doses of up to 120 mg
The core 36-week Phase 2b ACCESS study was a randomized, double-blind, placebo-controlled, Phase 2b dose-range finding clinical study that enrolled 230 adult participants living with obesity (body mass index (BMI) ≥ 30 kg/m2), or overweight (BMI ≥ 27 kg/m2) with at least one weight-related comorbidity. All participants were randomized 3:1 (active:placebo) and started at 5 mg of aleniglipron (or placebo) with a 4-week titration schedule, reaching target doses of 45 mg, 90 mg or 120 mg once-daily.
Each of the three doses in the ACCESS study achieved statistical significance on the primary endpoint and all key secondary endpoints. Primary efficacy estimandi results at 36 weeks are as follows:
|
Aleniglipron 45 mg |
Aleniglipron 90 mg |
Aleniglipron 120 mg |
Placebo | |
| Mean percent change in body weight at 36 weeks compared to baseline | -9.0 | -10.7 | -12.1 | -0.8 |
| Placebo-adjusted mean percent change in body weight at 36 weeks compared to baseline | -8.2 | -9.8 | -11.3 | - |
| P-value | p<0.0001 | p<0.0001 | p<0.0001 | - |
At Week 36, key secondary endpoints in the study show that 86% of participants in the aleniglipron 120 mg dose cohort achieved at least 5% weight loss and 70% achieved at least 10% weight loss. In addition, aleniglipron demonstrated clinically meaningful improvements in systolic blood pressure (-6.4 to -7.5 mmHg) and HbA1c (-0.28% to -0.37%).
Aleniglipron demonstrated a tolerability profile consistent with the GLP-1 receptor agonist class following repeated, once-daily dosing of up to 120 mg in the Phase 2b ACCESS study. As expected for the GLP1-RA drug class, the most common AEs were gastrointestinal (GI)-related and the two most common AEs in the titration phase were nausea and vomiting. AEs were generally observed early in treatment. In the Phase 2b ACCESS study, the AE-related treatment discontinuation rate ranged from 7.7% - 13.3% between all doses, with a mean 10.4% across all active arms in the study.
Exploratory ACCESS II Study - Evaluating higher doses up to 240 mg
ACCESS II is a randomized, double-blind, placebo-controlled, clinical study of aleniglipron that enrolled 85 adult participants living with obesity, or overweight with at least one weight-related comorbidity. The study was designed to evaluate two higher doses of aleniglipron. Participants started at 5 mg of aleniglipron (or placebo) and followed a 4-week titration schedule up to target doses of 120 mg, 180 mg and 240 mg. The 44-week study remains ongoing, with data from a prespecified 36-week analysis currently available.
At 36 weeks, each of the three dose cohorts in the ACCESS II study met statistical significance compared to placebo. Primary efficacy estimand results at 36 weeks are as follows:
|
Aleniglipron 120 mg |
Aleniglipron 180 mg |
Aleniglipron 240 mg |
Placebo | |
| Mean percent change in body weight at 36 weeks compared to baseline | -13.1 | -13.3 | -14.2 | +1.0 |
| Placebo-adjusted mean percent change in body weight at 36 weeks compared to baseline | -14.1 | -14.4 | -15.3 | - |
| P-value | p<0.0001 | p<0.0001 | p<0.0001 | - |
Aleniglipron demonstrated a tolerability profile consistent with the GLP-1 receptor agonist class following repeated, once- daily dosing of up to 240 mg. As expected for the GLP1-RA drug class, the most common AEs were GI-related and the two most common AEs in the titration phase were nausea and vomiting. AEs were generally observed early in treatment.
Body Composition Study - Evaluating lower 2.5 mg starting dose
Structure Therapeutics is currently conducting a randomized, placebo-controlled body composition study that enrolled 71 adult participants to assess the effect of aleniglipron (up to 120 mg) on body fat loss over a 40-week evaluation period. Participants in the aleniglipron treatment arm start at a 2.5 mg dose, and titrate up monthly to a target dose of 120 mg. Data from a pre-specified interim analysis after a median follow-up time of approximately 10 weeks showed that starting at a lower dose of 2.5 mg for the first four weeks meaningfully improved tolerability compared to what was observed at a starting titration dose of 5 mg in the ACCESS and ACCESS II studies, with no AE-related treatment discontinuations observed at the initial 2.5 mg dose or the subsequent 5 mg dose.
ACCESS Open-Label Extension Study - Following randomized 36-week period, evaluating lower 2.5mg starting dose
Following the 36-week randomized controlled portion of the Phase 2b ACCESS study, the majority of eligible ACCESS participants enrolled in ACCESS OLE. An initial analysis from the ongoing OLE demonstrates continuing weight loss in all dose cohorts out to 44 weeks, showing no evidence of weight loss plateau.
In the ACCESS OLE, participants who received placebo in the initial double-blind portion transitioned to aleniglipron at a starting dose of 2.5 mg and titrate monthly to a target dose of 120 mg. Initial data from this group of participants after eight weeks of treatment are consistent with the findings from the body composition study, showing that starting at a 2.5 mg titration dose meaningfully improved tolerability compared to what was observed in the starting 5 mg titration dose in ACCESS and ACCESS II studies, with no AE-related treatment discontinuations at the initial 2.5 mg or the subsequent 5 mg dose.
Aleniglipron Safety
Aleniglipron demonstrated a compelling safety profile across all studies. Importantly, there were no cases of drug-induced liver injury, no persistent liver enzyme elevations, and no QTc prolongation across all aleniglipron studies.
Phase 3 Preparation
Data from ACCESS, ACCESS II, body composition, and the ACCESS OLE studies provide a strong foundation to advance aleniglipron into Phase 3 clinical development. The Company plans to request a Type B End-of-Phase 2 meeting with the United States Food and Drug Administration (FDA) in the first half of 2026 to finalize the Phase 3 design, which is currently designed with a starting titration dose of 2.5 mg with the intent to evaluate multiple doses up to 240 mg. Structure Therapeutics anticipates initiating the Phase 3 program by mid-2026.
Conference Call and Webcast Information
Structure Therapeutics will host a conference call and webcast today, December 8, 2025 at 8:30 a.m. Eastern Time. A live webcast of the call will be available on the Investor Relations page of Structure Therapeutics’ website at https://ir.structuretx.com/events-presentations/events. To access the call by phone, participants should visit this link to receive dial-in details. The webcast will be made available for replay on Structure Therapeutics’ website beginning approximately two hours after the live event. The replay of the webcast will be available for 90 days.
About Aleniglipron and Structure Therapeutics’ Oral Metabolic Franchise
Aleniglipron (GSBR-1290) is an investigational orally-available, small molecule agonist of the glucagon-like-peptide-1 (GLP-1) receptor, a validated drug target for the treatment of obesity and type 2 diabetes mellitus (T2DM). Through Structure Therapeutics’ structure-based drug discovery platform, aleniglipron was designed to be a biased G Protein-Coupled Receptor (GPCR) agonist, which selectively activates the G-protein signaling pathway. Beyond aleniglipron, Structure Therapeutics is developing next generation oral small molecules including amylin receptor agonists, and other combination GLP-1 receptor agonists candidates such as glucose-dependent insulinotropic polypeptide (GIP), glucagon and apelin oral small molecules.
About Structure Therapeutics
Structure Therapeutics is a science-driven clinical-stage biopharmaceutical company focused on discovering and developing innovative
oral small molecule treatments for chronic metabolic conditions with significant unmet medical needs. Utilizing its next generation structure-based
drug discovery platform, the Company has established a robust GPCR-targeted pipeline, featuring multiple wholly-owned proprietary clinical-stage
oral small molecule compounds designed to surpass the scalability limitations of traditional biologic and peptide therapies and be accessible
to more people living with obesity around the world. For additional information, please visit www.structuretx.com.
Forward Looking Statements
This press release contains “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical fact are statements that could be deemed forward-looking statements, including, without limitation, statements concerning: the Company’s future plans and prospects; any expectations regarding the potential benefits, tolerability and safety profile, accessibility, scalability, combinability, capability, efficacy, convenience, expected effects and future application of aleniglipron; the belief that data to date from the Phase 2b ACCESS, Phase 2 ACCESS II, body composition, and the Phase 2b ACCESS OLE studies support and inform advancement of the Phase 3 clinical development of aleniglipron; the belief that aleniglipron represents a potentially best-in-class small molecule GLP1 and may be a backbone therapy for obesity; the expected timing for the meeting with the FDA to finalize the Phase 3 trial design and the Phase 3 program initiation of aleniglipron; any presumption that topline, interim or preliminary data will be representative of final data or data in later clinical trials; and the belief that the results from ACCESS program represent a promising advance in the therapeutic landscape and brings the Company closer to a future where people living with obesity have multiple, accessible options to address their needs. In addition, when or if used in this press release, the words and phrases “anticipated,” “believe,” “expect,” “may,” “on track,” “plan,” “potential,” “suggests,” “to be,” “to begin,” “will,” and similar expressions and their variants, as they relate to the Company may identify forward-looking statements. Forward-looking statements are neither historical facts nor assurances of future performance. Although the Company believes the expectations reflected in such forward-looking statements are reasonable, the Company can give no assurance that such expectations will prove to be correct. Readers are cautioned that actual results, levels of activity, safety, performance or events and circumstances could differ materially from those expressed or implied in the Company’s forward-looking statements due to a variety of risks and uncertainties, which include, without limitation: risks and uncertainties related to topline results that the Company reports are based on preliminary analysis of key efficacy and safety data, and such data may change following a more comprehensive review of the data related to the clinical trial and such topline data may not accurately reflect the complete results of a clinical trial, the preliminary nature of the results due to the length of the study and sample size and the results from earlier clinical studies not necessarily being predictive of future results; potential delays in the commencement, enrollment and completion of the Company’s planned clinical studies; the Company’s ability to advance aleniglipron, ACCG-2671, ACCG-3535, ANPA-0073, LTSE-2578, and its other therapeutic candidates, obtain regulatory approval of, and ultimately commercialize the Company’s therapeutic candidates; competitive products or approaches limiting the commercial value of the Company’s product candidates; the timing and results of preclinical and clinical studies; the Company’s ability to fund development activities and achieve development goals; the Company's reliance on third parties, including clinical research organizations, manufacturers, suppliers and collaborators, over which it may not always have full control; general geopolitical and macroeconomic conditions, including as a result of tariffs and various global conflicts; the Company’s ability to protect its intellectual property; and other risks and uncertainties described in the Company’s filings with the Securities and Exchange Commission (SEC), including the Company’s latest Quarterly Report on Form 10-Q and future reports the Company may file with the SEC from time to time. All forward-looking statements contained in this press release speak only as of the date on which they were made and are based on management’s assumptions and estimates as of such date. The Company undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made, except as required by law.
Investors:
Danielle Keatley
Structure Therapeutics Inc.
ir@structuretx.com
Media:
Dan Budwick
1AB
Dan@1abmedia.com
#####
i The primary efficacy estimand represents efficacy had all randomized participants remained on study treatment (with possible dose interruptions and/or dose modifications) for 36 weeks without initiating rescue weight management treatments or surgeries.
Exhibit 99.2

1 December 8, 2025 Aleniglipron Topline ACCESS Phase 2b Results

2 This presentation contains “forward - looking statements” within the meaning of the “safe harbor” provisions of the Private Securi ties Litigation Reform Act of 1995. All statements other than statements of historical fact are statements that could be deemed forward - looking statements, including, without limitation, statements concerning: the estimated addressable patient population, market, and revenue opportunity for aleniglipron; any expectations regarding the potential benefits, tolerability and safety profile, accessibility, dose flexibility, scalability, co st, combinability, capability, efficacy, convenience, expected effects, and future application of aleniglipron; the Company’s belief that data to date from the ACCESS, ACCESS II, Body Composition and Open Label Extension st udi es provide a strong foundation for the Phase 3 clinical development of aleniglipron; the belief that all key questions have been answered by the studies to date; the expected size and design for the Phase 3 trial; any presumption that topline, interim or preliminary data will be representative of final data or data in later clinical trials; the belief that the Company is well positioned to lead with a differentiated and highly scalable pi pel ine of oral small molecule medicines designed to address the significant unmet needs in obesity and related metabolic diseases; the belief that there is potential for expansion beyond obesity, including chronic kidney disease , m etabolic associated steatohepatitis, heart failure, sleep apnea, type 2 diabetes mellitus, osteoarthritis, and addiction; plans to conduct FDA regulatory interactions; the expected timing of topline data readouts fro m t he Open Label Extension, ACCESS II Extension, Body Composition, SWITCH and T2DM studies; the planned initiation of the ACCG - 3535 Phase 1 study and the timing thereof; the expected timing of study results from the Phase 1 ACCG - 2671 study; and the Company’s future plans and prospects. In addition, when or if used, the words and phrases “believe,” “may,” “potential,” “to be,” “will,” and similar expressions and their vari ant s, as they relate to the Company may identify forward - looking statements. Forward - looking statements are neither historical facts nor assurances of future performance. Although the Company believes the expectations ref lected in such forward - looking statements are reasonable, the Company can give no assurance that such expectations will prove to be correct. Readers are cautioned that actual results, levels of activity, saf ety , performance or events and circumstances could differ materially from those expressed or implied in the Company’s forward - looking statements due to a variety of risks and uncertainties, which include, without limitation: risk s and uncertainties related to topline results that the Company reports is based on preliminary analysis of key efficacy and safety data, and such data may change following a more comprehensive review of the data related to the clinical trial and such topline data may not accurately reflect the complete results of a clinical trial, the preliminary nature of the results due to the length of the study and sample size and results from earlier cl inical studies not necessarily being predictive of future results; potential delays in the commencement, enrollment and completion of the Company’s planned clinical studies; the Company’s ability to advance aleniglip ron , ACCG - 2671, ACCG - 3535, ANPA - 0073, LTSE - 2578, and its other therapeutic candidates, obtain regulatory approval of, and ultimately commercialize the Company’s therapeutic candidates; competitive pro duc ts or approaches limiting the commercial value of the Company’s product candidates; the timing and results of preclinical and clinical studies; the Company’s ability to fund development activities and achieve deve lop ment goals; the Company's reliance on third parties, including clinical research organizations, manufacturers, suppliers and collaborators, over which it may not always have full control; general geopolitical and macroeco nom ic conditions, including as a result of tariffs and various global conflicts; the Company’s ability to protect its intellectual property; and other risks and uncertainties described in the Company’s filings with the Securitie s a nd Exchange Commission (SEC), including the Company’s latest Quarterly Report on Form 10 - Q and future reports the Company may file with the SEC from time to time. All forward - looking statements contained in this present ation speak only as of the date on which they were made and are based on management’s assumptions and estimates as of such date. The Company undertakes no obligation to update such statements to reflect events t hat occur or circumstances that exist after the date on which they were made, except as required by law. This presentation discusses product candidates that are under clinical study and which have not yet been approved for marketi ng by the U.S. Food and Drug Administration. No representation is made as to the safety or effectiveness of these product candidates for the use for which such product candidates are being studied. This presentation may incorporate publicly - available third - party data that we have not independently verified. There are risks i nherent in conducting cross - trial comparisons and the results should be interpreted with caution. The presentation of such third - party data does not represent a head - to - head comparison of how our product candidates performed a gainst any other third - party product candidate or study. Rather, such third - party data has been pulled by us from publicly - available sources for supplemental informational purposes, only. We caution you that any compari sons against third - party data set forth herein should not be viewed as a side - by - side comparison, and you should not rely on the completeness or accuracy of our presentation of the results of any third - party drug c andidate in these slides, due to differences in study design, how other companies quantify or qualify eligibility criteria, and how results are recorded, among other distinguishing factors and uncertainties. Because we may be unaware of or may not adequately present various distinguishing factors and uncertainties, the comparisons set forth herein may not properly present such third - party data, which may differ materially from the data as pr esented here. Investors are encouraged to independently review third party data and should not rely on our presentation of such data as a single measure to evaluate our business. “Structure Therapeutics,” the Structure Therapeutics logo and other trademarks, trade names or service marks of the Company a ppe aring in this presentation are the property of the Company. All other trademarks, trade names and service marks appearing in this presentation are the property of their respective owners. Solely for convenience, t he trademarks and trade names in this presentation may be referred to without the ® and symbols, but such references should not be construed as any indicator that their respective owners will not assert their righ ts thereto. Forward - Looking Statements
3 Aleniglipron : A Backbone in Structure's Oral Small Molecule Portfolio Raymond Stevens Aleniglipron Phase 3 Readiness Blai Coll Topline 36 week Data from Phase 2b ACCESS and 3 Supplementary Studies Blai Coll, M.D., Ph.D, Chief Medical Officer Today’s Update Aleniglipron Obesity Opportunity Raymond Stevens, Ph.D, Chief Executive Officer 1 2 3 4 Q&A Blai Coll, Raymond Stevens Jun Yoon, Chief Financial Officer 5 ORAL SMALL MOLECULE GLP - 1 Receptor Agonist ALENIGLIPRON

4 Our Mission Making medicines more accessible to all

5 Aleniglipron Obesity Opportunity Raymond Stevens

6 1. August 2025 Symphony Health prescription data and 2025 Evaluate Pharma sales data 2. Trust for America’s Health Report https://www.tfah.org/report - details/stateofobesity2019/#:~:text=Obesity%20is%20a%20growing%20epidemic,100%20million%20people%20%E2%80%93%20have%20obes 3. Goldman Sachs Report https://www.goldmansachs.com/insights/articles/anti - obesity - drug - market 4. 2022 World Health Organization https://www.who.int/news - room/fact - sheets/detail/obesity - and - overweight 5. World Obesity Atlas 2024 https://s3 - eu - west - 1.amazonaws.com/wof - files/WOF_Obesity_Atlas_2024.pdf >5 million in US 1 1.5 billion people in 2030 5 >$100 billion Total Addressable Market 3 (obesity or overweight with at least one weight - related comorbid condition) >800 million worldwide 4 >100 million in US 2 Overweight & Obesity Current Injectable Peptide GLP - 1s <5% of addressable market Access to Obesity Treatments Remains a Worldwide Unmet Need Only oral small molecules can scale to meet the needs of the global obesity patient population Overweight & Obesity
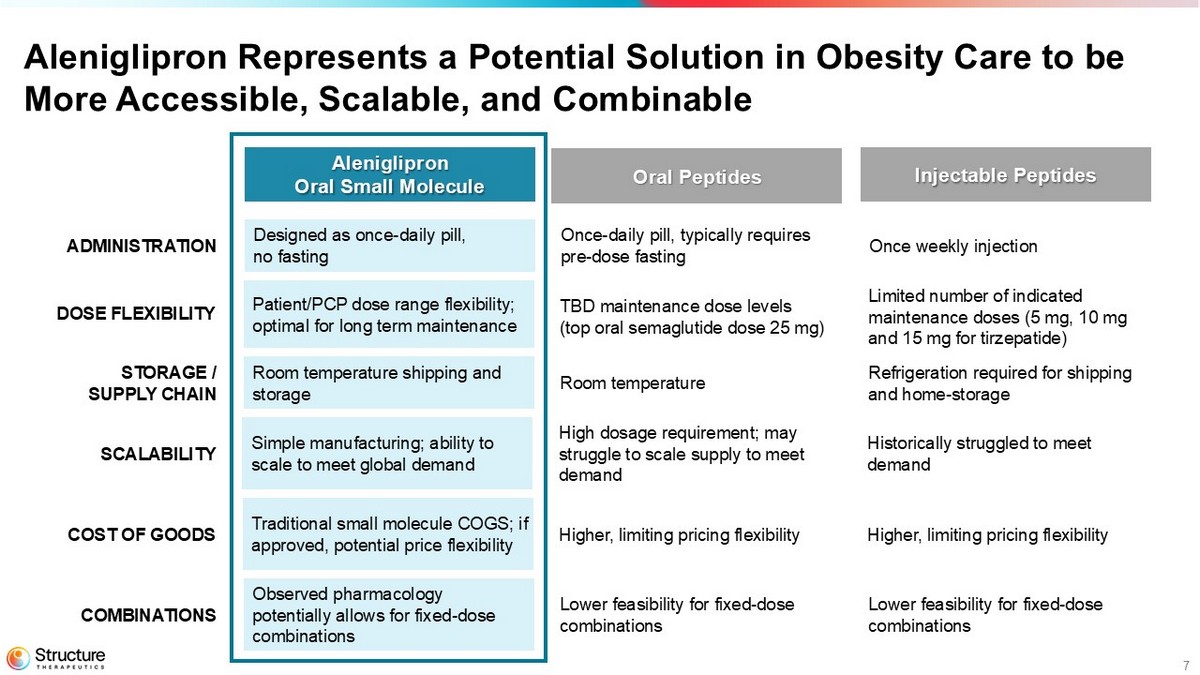
7 Aleniglipron Represents a Potential Solution in Obesity Care to be More Accessible, Scalable, and Combinable Aleniglipron Oral Small Molecule Oral Peptides Injectable Peptides ADMINISTRATION Designed as o nce - daily pill, no fasting Once - daily pill, typically requires pre - dose fasting Once weekly injection COMBINATIONS Observed p harmacology potentially allows for fixed - dose combinations Lower feasibility for fixed - dose combinations Lower feasibility for fixed - dose combinations SCALABILITY Simple manufacturing; ability to scale to meet global demand High dosage requirement; may struggle to scale supply to meet demand Historically struggled to meet demand COST OF GOODS Traditional small molecule COGS ; if approved, potential price flexibility Higher, limiting pricing flexibility Higher, limiting pricing flexibility Room temperature shipping and storage Room temperature Refrigeration required for shipping and home - storage STORAGE / SUPPLY CHAIN DOSE FLEXIBILITY Patient/PCP dose range flexibility; o ptimal for long term maintenance TBD maintenance dose levels (top oral semaglutide dose 25 mg) Limited number of indicated maintenance doses (5 mg, 10 mg and 15 mg for tirzepatide)

8 Aleniglipron Obesity Data Summary Differentiated Selective Oral Small Molecule GLP - 1R Receptor Agonist Safety • > 500 participants treated across all studies up to 44 weeks o No events of drug induced liver injury o No off - target safety signals across all dose levels o No events of QTc prolongation Tolerability • Phase 2b ACCESS : o GI - related AEs consistent with GLP - 1RA class o Overall 10.4% AE - related treatment discontinuations • Exploratory ACCESS II : o For those participants who achieved re - randomization – No AE - related treatment discontinuations up to 240 mg dose • Open Label Extension (OLE) & Body Composition: o Clinically meaningful improvement in tolerability when starting at 2.5 mg compared to 5 mg start o No discontinuations observed to date* Efficacy • Phase 2b ACCESS 36 week placebo - adjusted mean weight loss: o 8.2% at 45 mg o 9.8% at 90 mg o 11.3% at 120 mg • Exploratory ACCESS II 36 week p lacebo - adjusted mean weight loss: o 14.1% at 120 mg o 14.4% at 180 mg o 15.3% at 240 mg • No evidence of weight loss plateau • Proportional pharmacokinetic (PK) exposure up to 240 mg • Clinically meaningful improvements in blood pressure and HbA1c *Study ongoing and interim data from a pre - specified analysis. Interim data as of November 25, 2025

9 Topline 36 Week Data Phase 2b ACCESS and Three Supplementary Studies Blai Coll
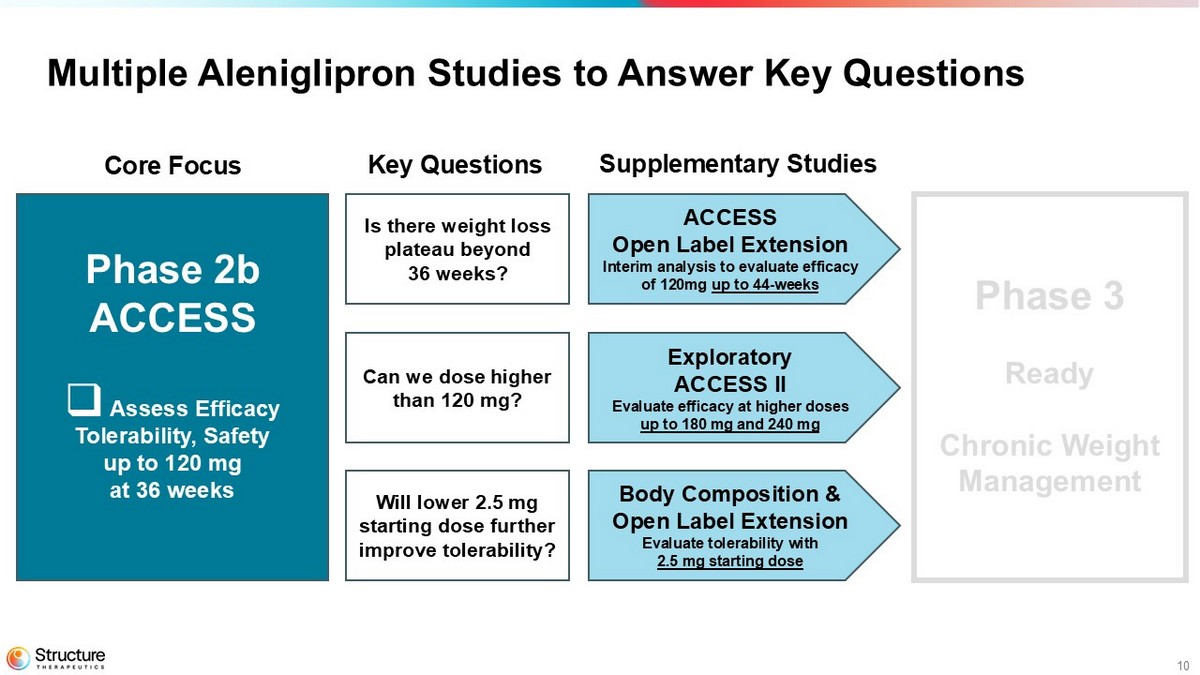
10 Multiple Aleniglipron Studies to Answer Key Questions Phase 2b ACCESS □ Assess Efficacy Tolerability, Safety up to 120 mg at 36 weeks Phase 3 Ready Chronic Weight Management ACCESS Open Label Extension Interim analysis to evaluate efficacy of 120mg up to 44 weeks Exploratory ACCESS II Evaluate efficacy at higher doses up to 180 mg and 240 mg Body Composition & Open Label Extension Evaluate tolerability with 2.5 mg starting dose Core Focus Key Questions Supplementary Studies Is there weight loss plateau beyond 36 weeks? Can we dose higher than 120 mg? Will lower 2.5 mg starting dose further improve tolerability?
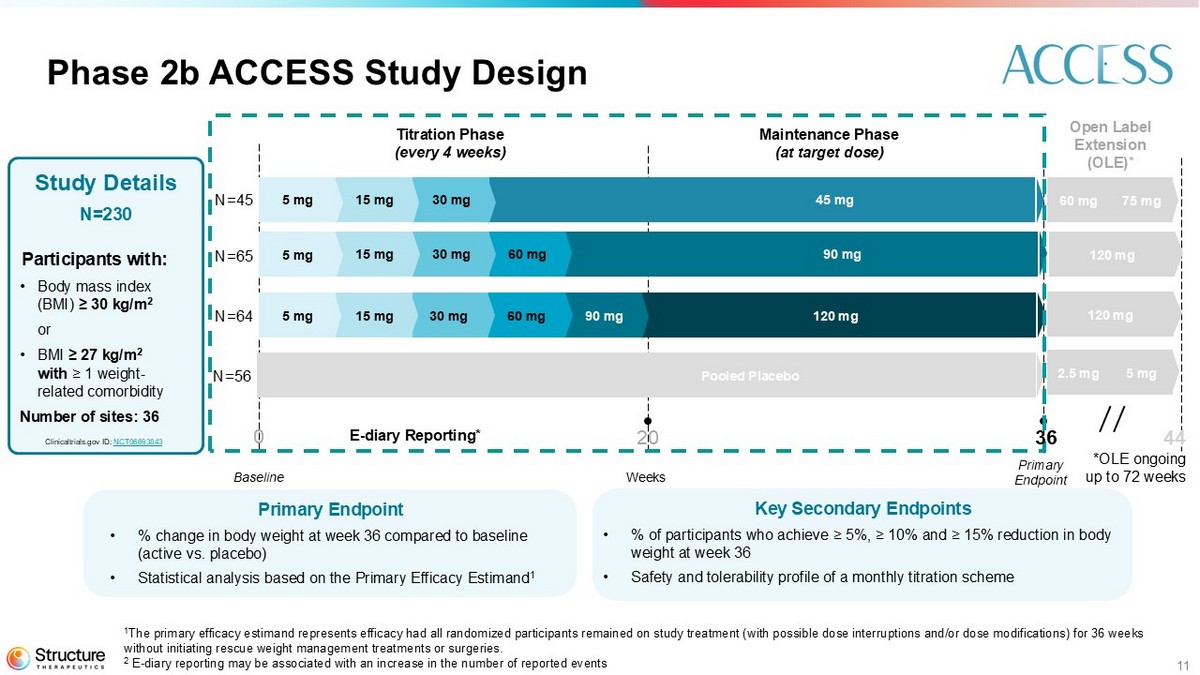
11 Phase 2b ACCESS Study Design Key S econdary Endpoints • % of participants who achieve ≥ 5%, ≥ 10% and ≥ 15% reduction in body weight at week 36 • Safety and tolerability profile of a monthly titration scheme Primary Endpoint • % change in body weight at week 36 compared to baseline (active vs. placebo) • Statistical analysis based on the Primary Efficacy Estimand 1 Study Details N=230 Participants with: • Body mass index (BMI) ≥ 30 kg/m 2 or • BMI ≥ 27 kg/m 2 with ≥ 1 weight - related comorbidity Number of sites: 36 Weeks Baseline Titration Phase (every 4 weeks) Maintenance Phase (at target dose) Primary Endpoint Clinicaltrials.gov ID: NCT06693843 Open Label Extension (OLE)* 90 mg 45 mg 60 mg 30 mg 15 mg 5 mg 30 mg 15 mg 5 mg 120 mg 90 mg 60 mg 30 mg 15 mg 5 mg *OLE ongoing up to 72 weeks 75 mg 60 mg Pooled Placebo 5 mg 2.5 mg 120 mg 120 mg E - diary Reporting* 0 20 36 44 N=45 N=65 N=64 N=56 1 The primary efficacy estimand represents efficacy had all randomized participants remained on study treatment (with possible dos e interruptions and/or dose modifications) for 36 weeks without initiating rescue weight management treatments or surgeries. 2 E - diary reporting may be associated with an increase in the number of reported events
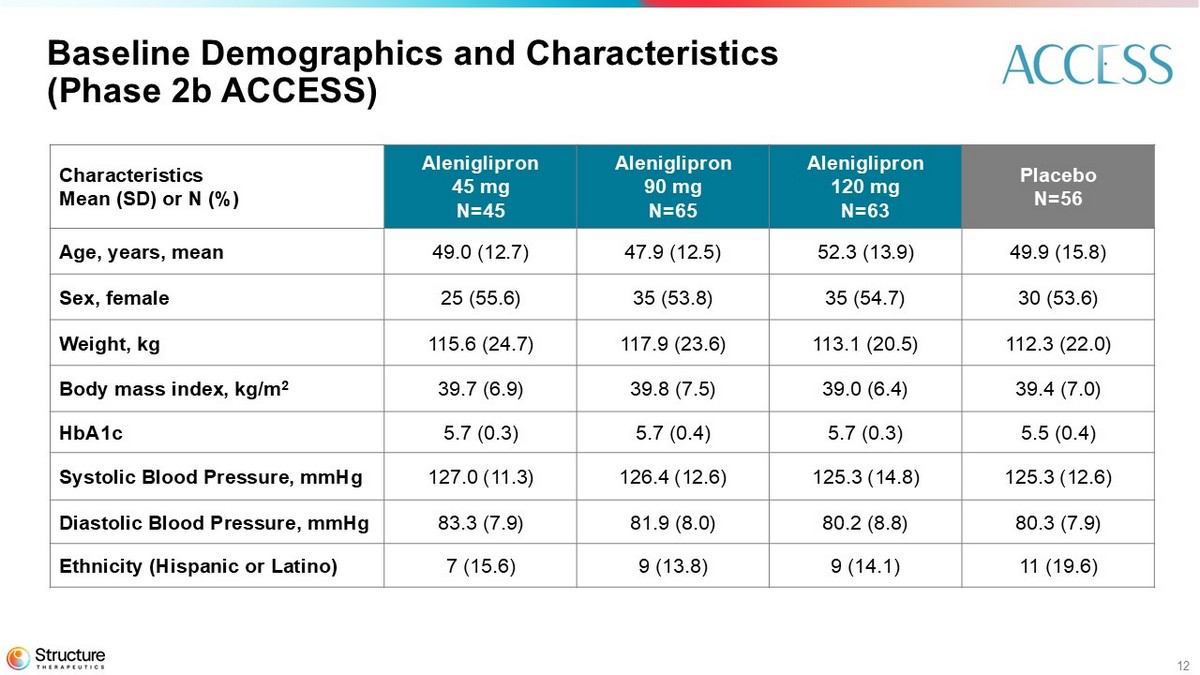
12 Baseline Demographics and Characteristics (Phase 2b ACCESS) Placebo N=56 Aleniglipron 120 mg N=63 Aleniglipron 90 mg N=65 Aleniglipron 45 mg N=45 Characteristics Mean (SD) or N (%) 49.9 (15.8) 52.3 (13.9) 47.9 (12.5) 49.0 (12.7) Age, years, mean 30 (53.6) 35 (54.7) 35 (53.8) 25 (55.6) Sex, female 112.3 (22.0) 113.1 (20.5) 117.9 (23.6) 115.6 (24.7) Weight, kg 39.4 (7.0) 39.0 (6.4) 39.8 (7.5) 39.7 (6.9) Body mass index, kg/m 2 5.5 (0.4) 5.7 (0.3) 5.7 (0.4) 5.7 (0.3) HbA1c 125.3 (12.6) 125.3 (14.8) 126.4 (12.6) 127.0 (11.3) Systolic Blood Pressure, mmHg 80.3 (7.9) 80.2 (8.8) 81.9 (8.0) 83.3 (7.9) Diastolic Blood Pressure, mmHg 11 (19.6) 9 (14.1) 9 (13.8) 7 (15.6) Ethnicity (Hispanic or Latino)
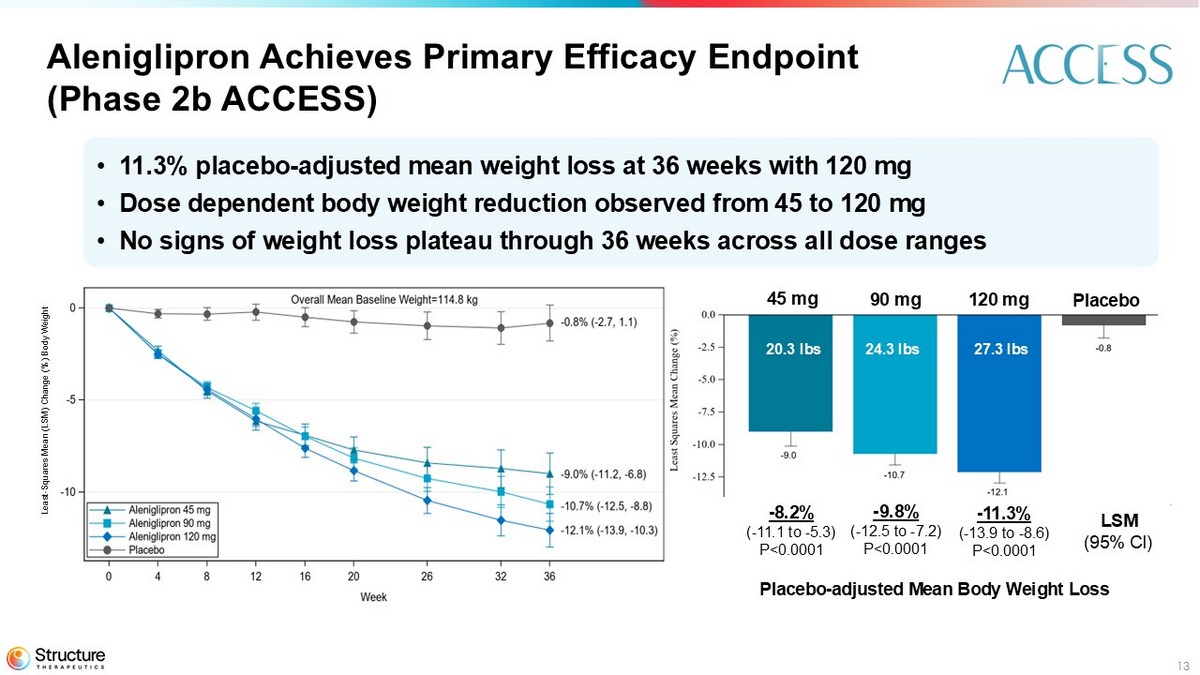
13 • 11.3% placebo - adjusted mean weight loss at 36 weeks with 120 mg • Dose dependent body weight reduction observed from 45 to 120 mg • No signs of weight loss plateau through 36 weeks across all dose ranges Aleniglipron Achieves Primary Efficacy Endpoint (Phase 2b ACCESS) 27.3 lbs 24.3 lbs 20.3 lbs - 8.2% ( - 11.1 to - 5.3) P<0.0001 - 9.8% ( - 12.5 to - 7.2) P<0.0001 - 11.3% ( - 13.9 to - 8.6) P<0.0001 LSM (95% CI) 45 mg 90 mg 120 mg Placebo Least - Squares Mean (LSM) Change (%) Body Weight
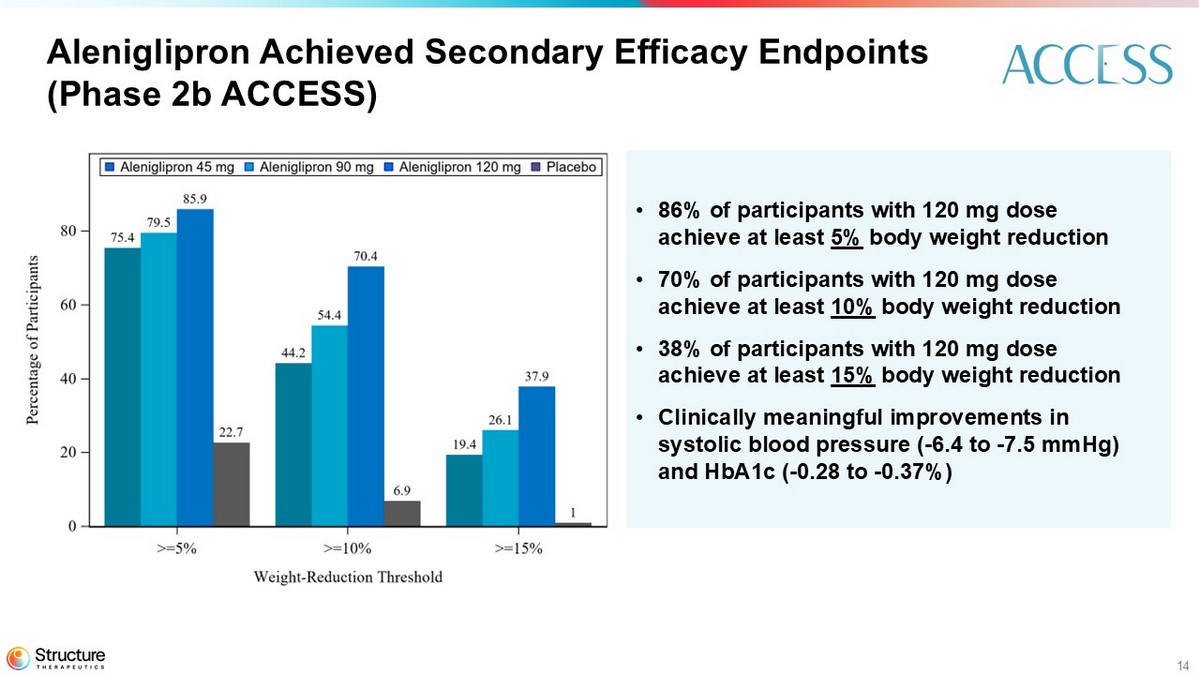
14 Aleniglipron Achieved Secondary Efficacy Endpoints (Phase 2b ACCESS) • 86% of participants with 120 mg dose achieve at least 5% body weight reduction • 70% of participants with 120 mg dose achieve at least 10% body weight reduction • 38% of participants with 120 mg dose achieve at least 15% body weight reduction • Clinically meaningful improvements in systolic blood pressure ( - 6.4 to - 7.5 mmHg) and HbA1c ( - 0.28 to - 0.37%)
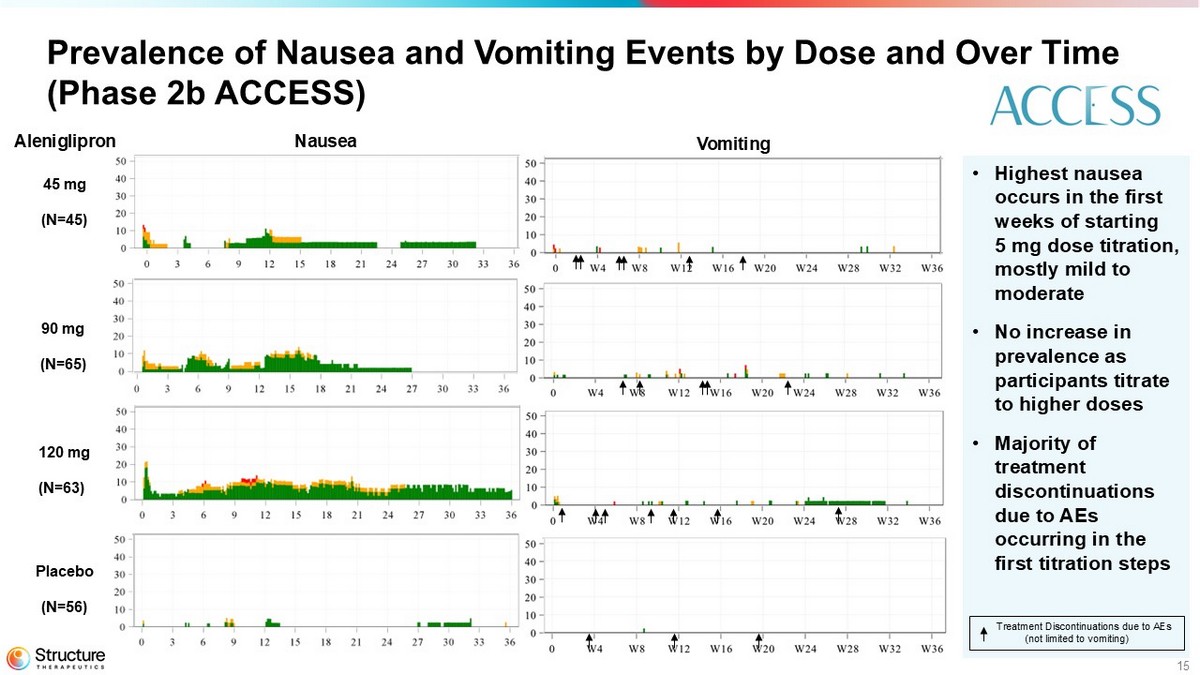
15 Prevalence of Nausea and Vomiting Events by Dose and Over Time (Phase 2b ACCESS) 45 mg (N=45) Placebo (N=56) 120 mg (N=63) 90 mg (N=65) • Highest nausea occurs in the first weeks of starting 5 mg dose titration, mostly mild to moderate • No increase in prevalence as participants titrate to higher doses • Majority of treatment discontinuations due to AEs occurring in the first titration steps Treatment Discontinuations due to AEs (not limited to vomiting) Nausea Vomiting Aleniglipron
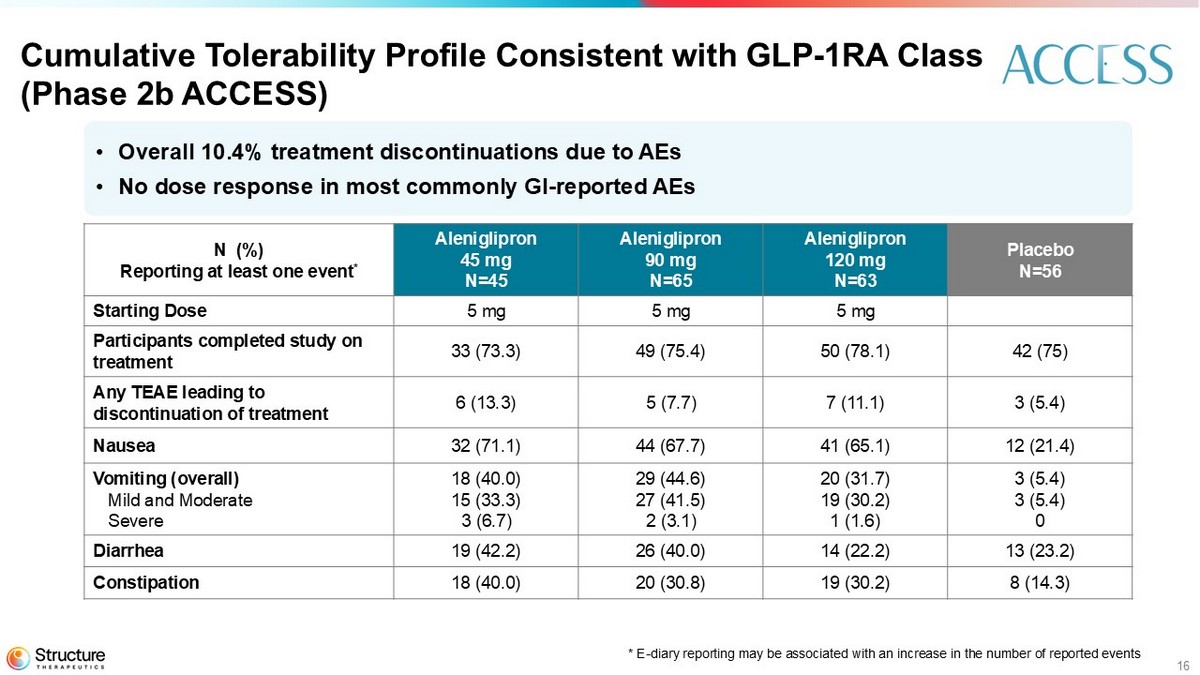
16 Cumulative Tolerability Profile Consistent with GLP - 1RA Class (Phase 2b ACCESS) Placebo N=56 Aleniglipron 120 mg N=63 Aleniglipron 90 mg N=65 Aleniglipron 45 mg N=45 N (%) Reporting at least one event * 5 mg 5 mg 5 mg Starting Dose 42 (75) 50 (78.1) 49 (75.4) 33 (73.3) Participants completed study on treatment 3 (5.4) 7 (11.1) 5 (7.7) 6 (13.3) Any TEAE leading to discontinuation of treatment 12 (21.4) 41 (65.1) 44 (67.7) 32 (71.1) Nausea 3 (5.4) 3 (5.4) 0 20 (31.7) 19 (30.2) 1 (1.6) 29 (44.6) 27 (41.5) 2 (3.1) 18 (40.0) 15 (33.3) 3 (6.7) Vomiting (overall) Mild and Moderate Severe 13 (23.2) 14 (22.2) 26 (40.0) 19 (42.2) Diarrhea 8 (14.3) 19 (30.2) 20 (30.8) 18 (40.0) Constipation • Overall 10.4% treatment discontinuations due to AEs • No dose response in most commonly GI - reported AEs *E - diary reporting is associated with an increase in the number of reported events

17 • 36 week placebo - adjusted mean weight loss : o 8.2% at 45 mg o 9.8% at 90 mg o 11.3% at 120 mg • No signs of weight loss plateau • 4 week titration to optimize tolerability: o GI - related AEs consistent with GLP - 1RA class o Overall 10.4% AE - related treatment discontinuations Summary Aleniglipron Topline Phase 2b ACCESS Results Phase 2b ACCESS Assess Efficacy Tolerability, Safety up to 120 mg at 36 weeks Core Focus
18 Multiple Aleniglipron Studies to Answer Key Questions Phase 2b ACCESS □ Assess Efficacy Tolerability, Safety up to 120 mg at 36 weeks Phase 3 Chronic Weight Management ACCESS Open Label Extension Interim analysis to evaluate efficacy of 120mg up to 44 - weeks Exploratory ACCESS II Evaluate efficacy at higher doses up to 180 mg and 240 mg Body Composition & Open Label Extension Evaluate tolerability with 2.5 mg starting dose Core Focus Key Questions Supplementary Studies Is there weight loss plateau beyond 36 weeks? Can we dose higher than 120 mg? Will lower 2.5 mg starting dose further improve tolerability?
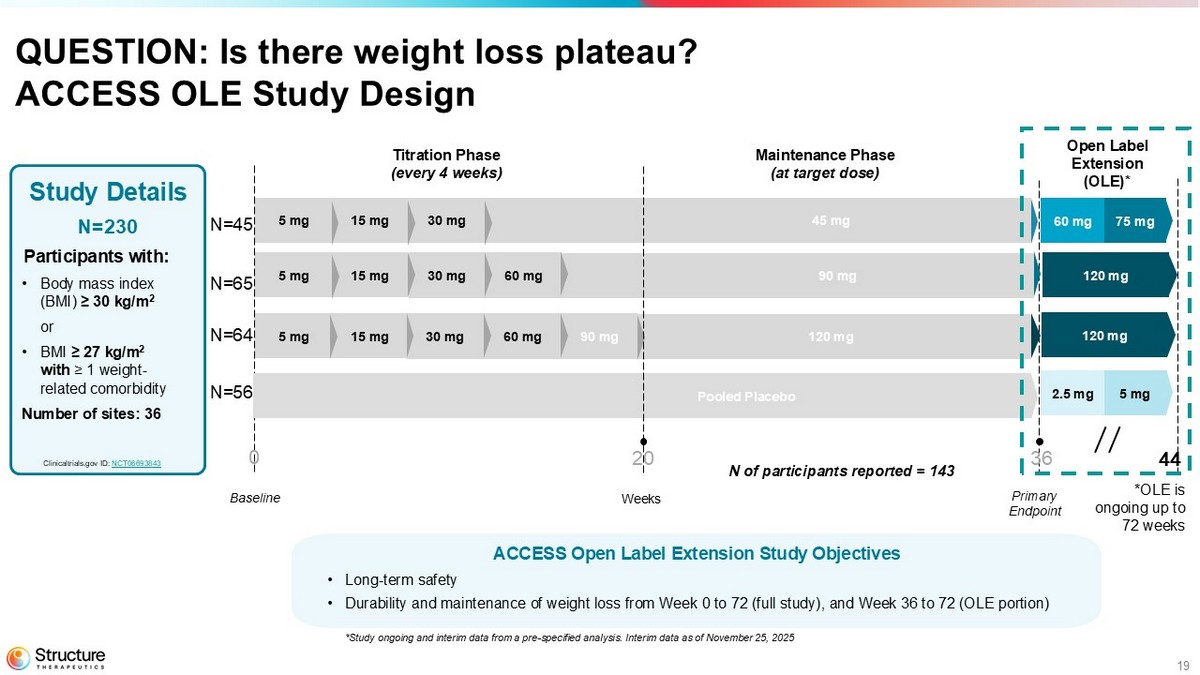
19 QUESTION: Is there weight loss plateau? ACCESS OLE Study Design Study Details N=230 Participants with: • Body mass index (BMI) ≥ 30 kg/m 2 or • BMI ≥ 27 kg/m 2 with ≥ 1 weight - related comorbidity Number of sites: 36 Weeks Baseline Titration Phase (every 4 weeks) Maintenance Phase (at target dose) Primary Endpoint Clinicaltrials.gov ID: NCT06693843 90 mg 45 mg 60 mg 30 mg 15 mg 5 mg 30 mg 15 mg 5 mg 120 mg 0 20 36 90 mg 60 mg 30 mg 15 mg 5 mg 44 N=45 N=65 N=64 N=56 75 mg 60 mg Pooled Placebo 5 mg 2.5 mg 120 mg 120 mg *OLE is ongoing up to 72 weeks Open Label Extension (OLE)* N of participants reported = 143 ACCESS Open Label Extension Study Objectives • Long - term safety • Durability and maintenance of weight loss from Week 0 to 72 (full study), and Week 36 to 72 (OLE portion) *Study ongoing and interim data from a pre - specified analysis. Interim data as of November 25, 2025
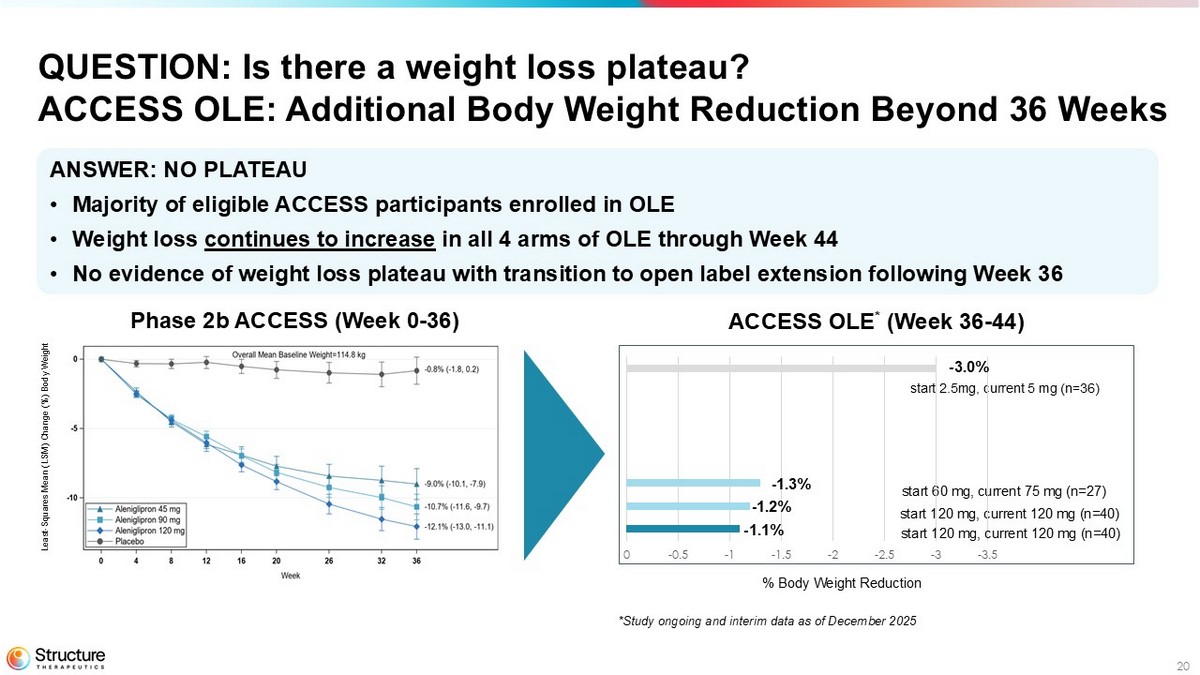
20 Phase 2b ACCESS (Week 0 - 36) ACCESS OLE * (Week 36 - 44) ANSWER: NO PLATEAU • Majority of eligible ACCESS participants enrolled in OLE • Weight loss continues to increase in all 4 arms of OLE through Week 44 • No evidence of weight loss plateau with transition to open label extension following Week 36 QUESTION: Is there a weight loss plateau? ACCESS OLE: Additional Body Weight Reduction Beyond 36 Weeks % Body Weight Reduction start 2.5mg, current 5 mg (n=36) start 60 mg, current 75 mg (n=27) start 120 mg, current 120 mg (n=40) -3.5 -3 -2.5 -2 -1.5 -1 -0.5 0 start 120 mg, current 120 mg (n=40) - 3.0% - 1.3% - 1.2% - 1.1% *Study ongoing and interim data as of December 2025 Least - Squares Mean (LSM) Change (%) Body Weight
21 Multiple Aleniglipron Studies to Answer Key Questions Phase 2b ACCESS □ Assess Efficacy Tolerability, Safety up to 120 mg at 36 weeks Phase 3 Chronic Weight Management ACCESS Open Label Extension Interim analysis to evaluate efficacy of 120mg up to 44 - weeks Exploratory ACCESS II Evaluate efficacy at higher doses up to 180 mg and 240 mg Body Composition & Open Label Extension Evaluate tolerability with 2.5 mg starting dose Core Focus Key Questions Supplementary Studies Can we dose higher than 120 mg? Will lower 2.5 mg starting dose further improve tolerability? Is there weight loss plateau beyond 36 weeks?
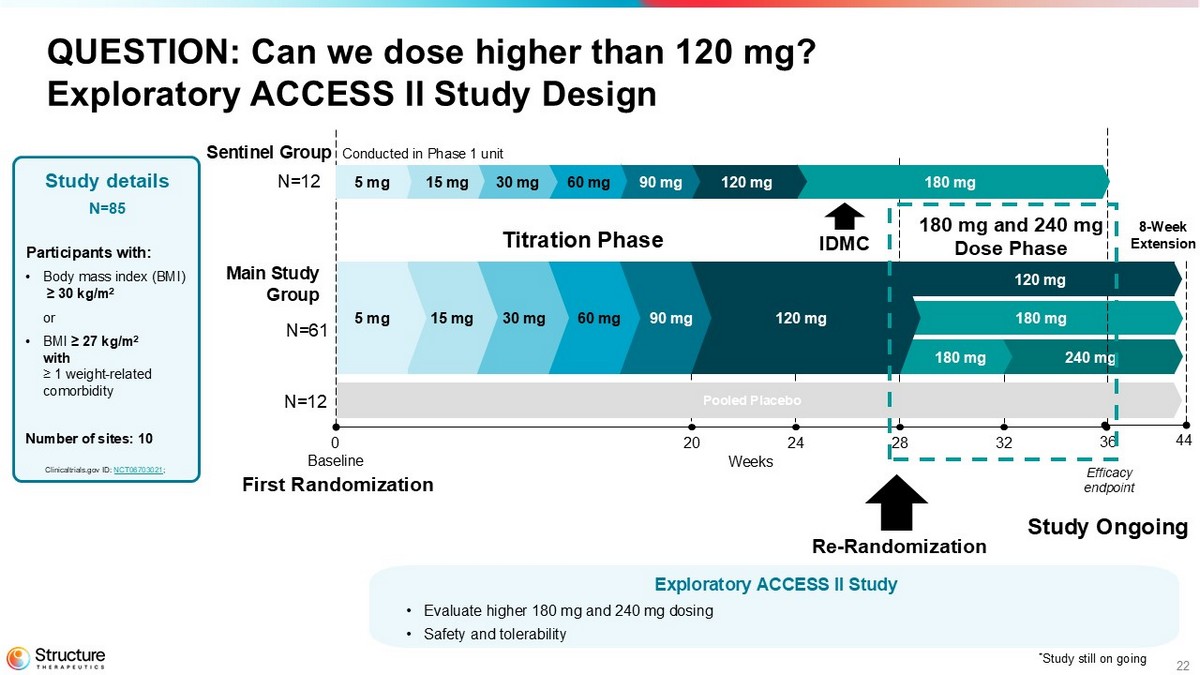
22 240 mg 180 mg QUESTION: Can we dose higher than 120 mg? Exploratory ACCESS II Study Design Weeks 0 20 24 28 32 36 Baseline 180 mg 120 mg 90 mg 60 mg 30 mg 15 mg 5 mg Study details N=85 Participants with: • Body mass index (BMI) ≥ 30 kg/m 2 or • BMI ≥ 27 kg/m 2 with ≥ 1 weight - related comorbidity Number of sites: 10 Clinicaltrials.gov ID: NCT06703021 ; 120 mg 8 - Week Extension 44 Efficacy endpoint Re - Randomization N=61 180 mg Sentinel Group 120 mg 90 mg 60 mg 30 mg 15 mg 5 mg Main Study Group N=12 IDMC Titration Phase 180 mg and 240 mg Dose Phase * Study still on going Pooled Placebo N=12 First Randomization Exploratory ACCESS II Study • Evaluate higher 180 mg and 240 mg dosing • Safety and tolerability Study Ongoing Conducted in Phase 1 unit
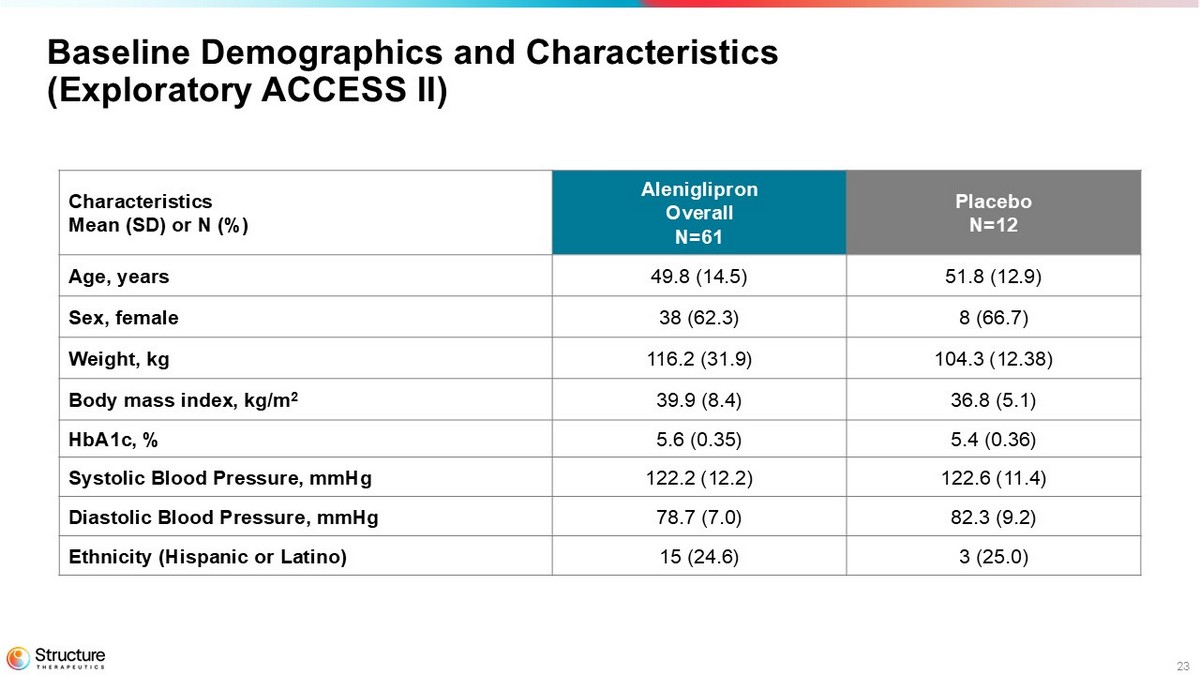
23 Placebo N=12 Aleniglipron Overall N=61 Characteristics Mean (SD) or N (%) 51.8 (12.9) 49.8 (14.5) Age, years 8 (66.7) 38 (62.3) Sex, female 104.3 (12.38) 116.2 (31.9) Weight, kg 36.8 (5.1) 39.9 (8.4) Body mass index, kg/m 2 5.4 (0.36) 5.6 (0.35) HbA1c, % 122.6 (11.4) 122.2 (12.2) Systolic Blood Pressure, mmHg 82.3 (9.2) 78.7 (7.0) Diastolic Blood Pressure, mmHg 3 (25.0) 15 (24.6) Ethnicity (Hispanic or Latino) Baseline Demographics and Characteristics (Exploratory ACCESS II)
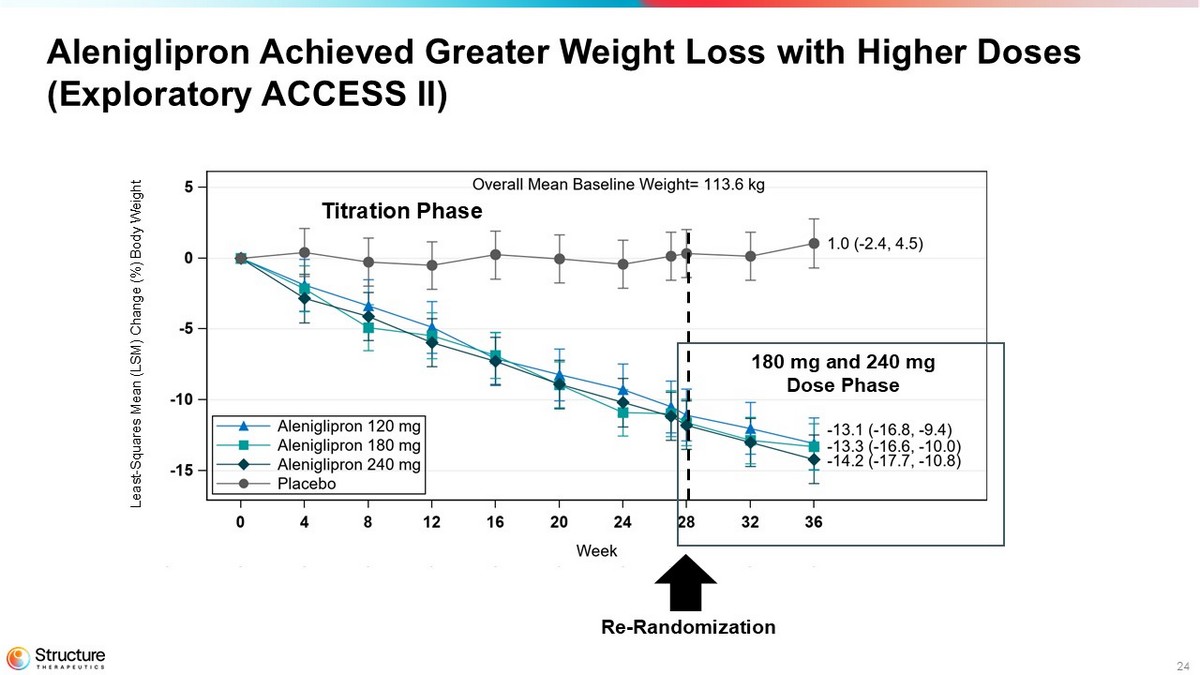
24 Aleniglipron Achieved Greater Weight Loss with Higher Doses (Exploratory ACCESS II) Re - Randomization Titration Phase 180 mg and 240 mg Dose Phase Least - Squares Mean (LSM) Change (%) Body Weight
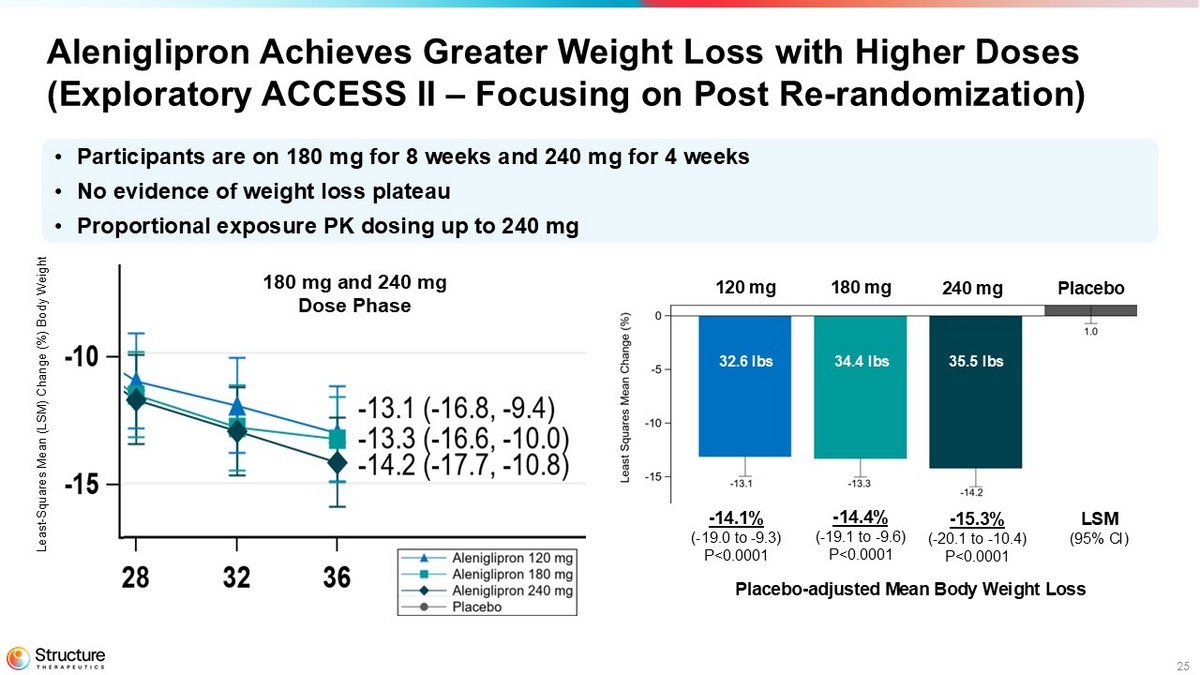
25 Aleniglipron Achieves Greater Weight Loss with Higher Doses (Exploratory ACCESS II – Focusing on Post Re - randomization) Analysis on going - draft results - 14.1% ( - 19.0 to - 9.3) P<0.0001 180 mg and 240 mg Dose Phase - 14.4% ( - 19.1 to - 9.6) P<0.0001 - 15.3% ( - 20.1 to - 10.4) P<0.0001 LSM (95% CI) 120 mg 180 mg 240 mg Placebo • Participants are on 180 mg for 8 weeks and 240 mg for 4 weeks • No evidence of weight loss plateau • Proportional exposure PK dosing up to 240 mg Least - Squares Mean (LSM) Change (%) Body Weight 32.6 lbs 34.4 lbs 35.5 lbs
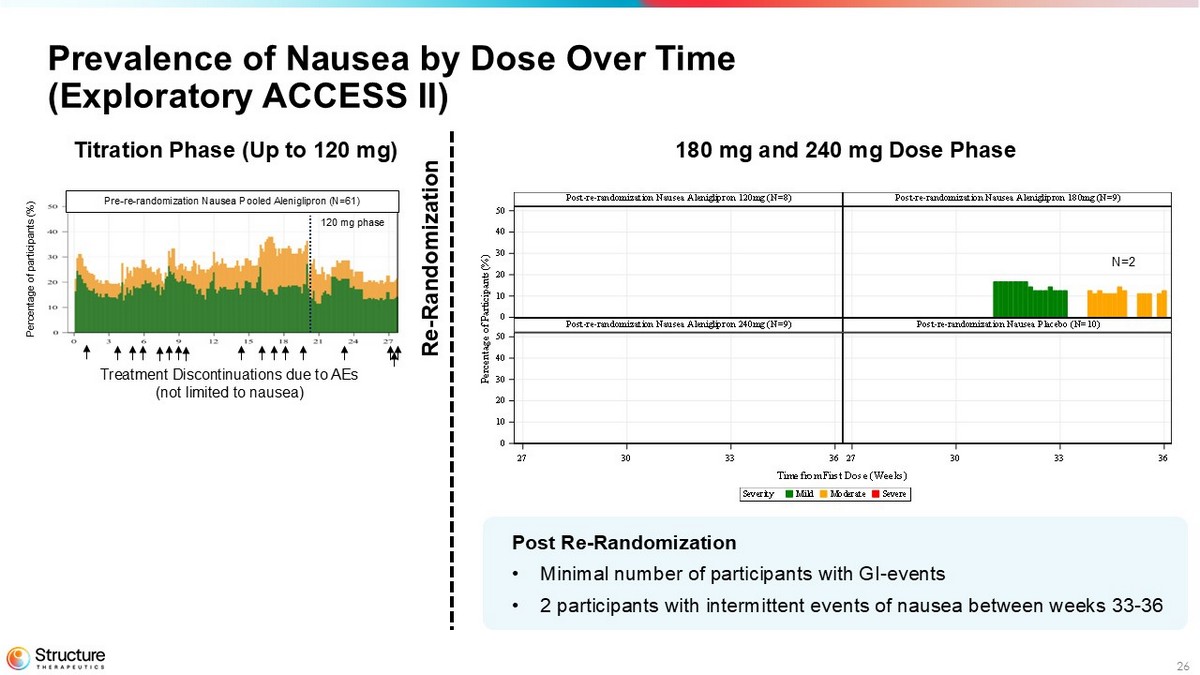
26 Prevalence of Nausea by Dose Over Time (Exploratory ACCESS II) Post Re - Randomization • Minimal number of participants with GI - events • 2 participants with intermittent events of nausea between weeks 33 - 36 SevereModerateMildSeverity Time from First Dose (Weeks) P e r c e n t a g e o f P a r t i c i p a n t s ( % ) Post-re-randomization Nausea Placebo (N=10)Post-re-randomization Nausea Aleniglipron 240mg (N=9) Post-re-randomization Nausea Aleniglipron 180mg (N=9)Post-re-randomization Nausea Aleniglipron 120mg (N=8) 27 30 33 3627 30 33 36 0 10 20 30 40 50 0 10 20 30 40 50 N=2 180 mg and 240 mg Dose Phase Titration Phase (Up to 120 mg) Pre - re - randomization Nausea Pooled Aleniglipron (N=61) Percentage of participants (%) Treatment Discontinuations due to AEs (not limited to nausea) 120 mg phase Re - Randomization
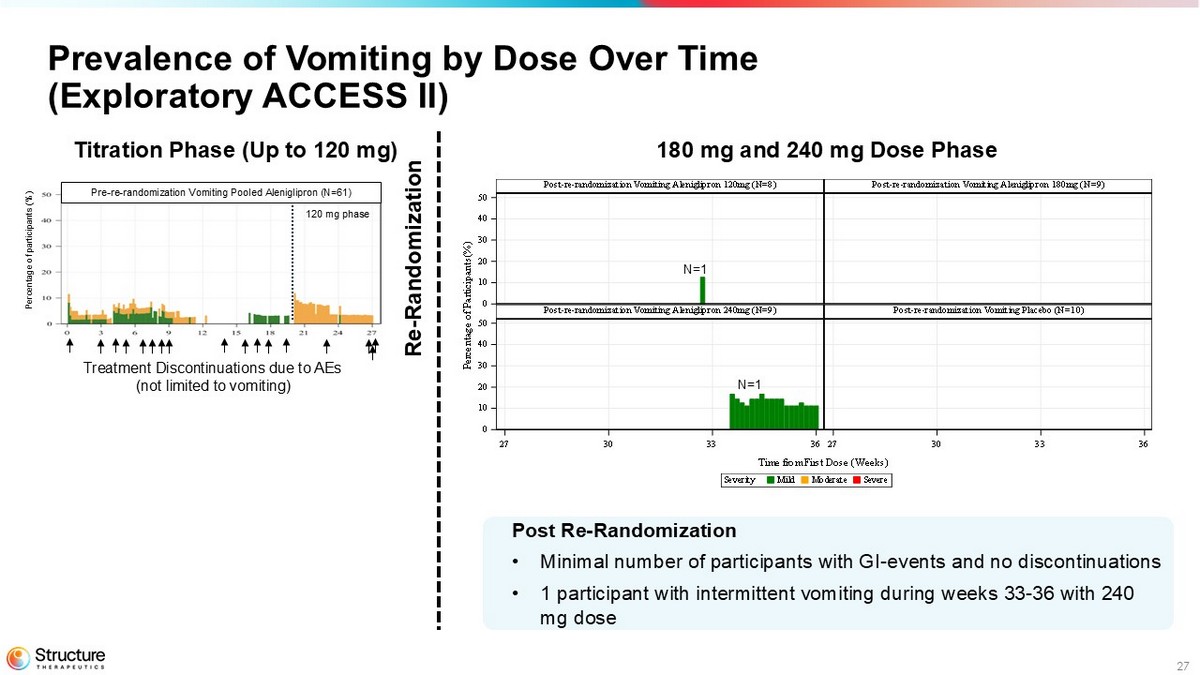
27 SevereModerateMildSeverity Time from First Dose (Weeks) P e r c e n t a g e o f P a r t i c i p a n t s ( % ) Post-re-randomization Vomiting Placebo (N=10)Post-re-randomization Vomiting Aleniglipron 240mg (N=9) Post-re-randomization Vomiting Aleniglipron 180mg (N=9)Post-re-randomization Vomiting Aleniglipron 120mg (N=8) 27 30 33 3627 30 33 36 0 10 20 30 40 50 0 10 20 30 40 50 Prevalence of Vomiting by Dose Over Time (Exploratory ACCESS II) N=1 N=1 180 mg and 240 mg Dose Phase Pre - re - randomization Vomiting Pooled Aleniglipron (N=61) Percentage of participants (%) Treatment Discontinuations due to AEs (not limited to vomiting) 120 mg phase Post Re - Randomization • Minimal number of participants with GI - events and no discontinuations • 1 participant with intermittent vomiting during weeks 33 - 36 with 240 mg dose Re - Randomization Titration Phase (Up to 120 mg)

28 Cumulative Tolerability Results Consistent with GLP - 1RA Class (Exploratory ACCESS II) 180 mg and 240mg Dosing Phase (Re - randomized Study Ongoing) Titration Phase (Up to 120 mg) Placebo N=10 Aleniglipron 120 mg to 240 mg N=26 Placebo N=12 Aleniglipron Up to 120 mg N=61 N (%) Reporting at least one event * 120/180 mg 5 mg Starting Dose 0 0 0 17 (27.9) Any TEAE leading to Treatment Discontinuation 0 0 0 11 (18) Non - AE related Treatment Discontinuation 0 0 1 (8.3) 6 (9.8) Non Re - randomized Participants in the Study 0 3 (11.5) 0 42 (68.9) Nausea 0 0 0 3 (11.5) 3 (11.5) 0 1 (8.3) 1 (8.3) 0 27 (44.3) 26 (42.6) 1 (1.6) Vomiting (Overall) Mild and Moderate Severe 0 0 1 (8.3) 22 (36.1) Diarrhea 0 0 2 (16.7) 13 (21.3) Constipation N o discontinuations at 120, 180, or 240 mg dose post r e - randomization at 36 weeks; study ongoing to 44 weeks *E - diary reporting is associated with an increase in the number of reported events

29 ANSWER: Additional activity and weight loss directionality confirmed up to 180 and 240 mg • Directionality in efficacy (placebo - adjusted mean weight loss, 36 week ) o 14.1% at 120 mg o 14.4% at 180 mg o 15.3% at 240 mg • Proportionality in exposure • Tolerability o For those participants who achieved re - randomization – No AE - related treatment discontinuations up to 240 mg dose at week 36; study ongoing to week 44 • Supports dose optimization strategy for Phase 3 Summary Aleniglipron ACCESS II Interim Results Exploratory ACCESS II Evaluate efficacy at higher doses up to 180 mg and 240 mg Can we dose higher than 120 mg?
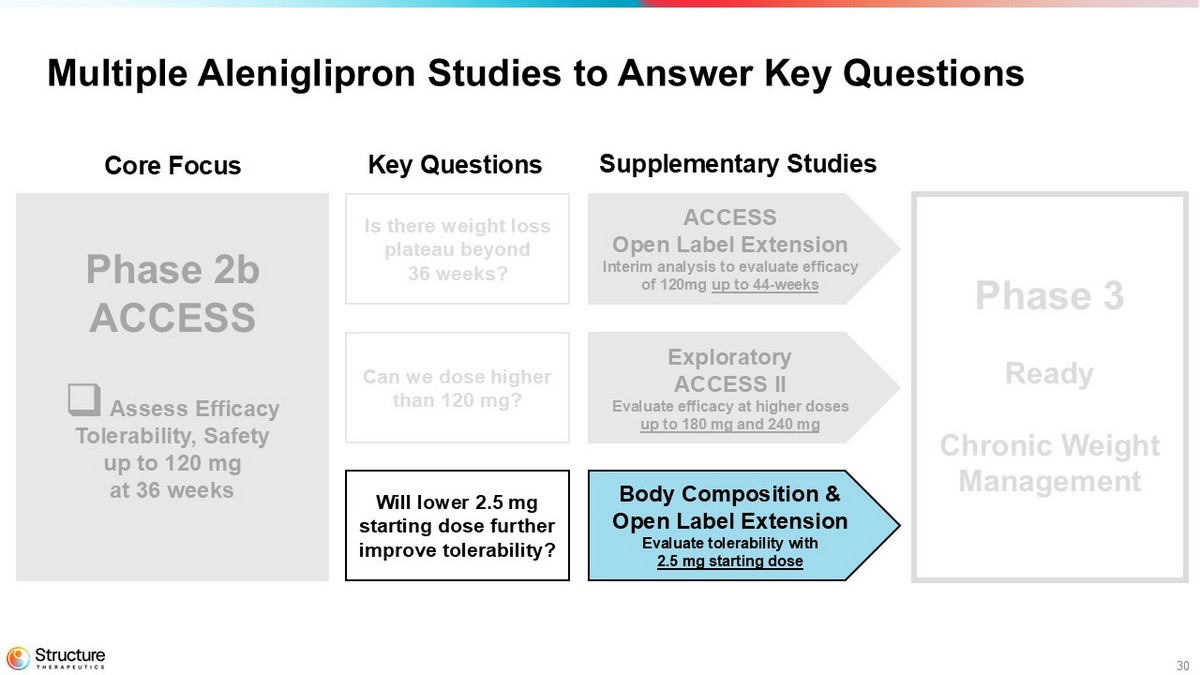
30 Multiple Aleniglipron Studies to Answer Key Questions Phase 2b ACCESS □ Assess Efficacy Tolerability, Safety up to 120 mg at 36 weeks Phase 3 Chronic Weight Management ACCESS Open Label Extension Interim analysis to evaluate efficacy of 120mg up to 44 - weeks Exploratory ACCESS II Evaluate efficacy at higher doses up to 180 mg and 240 mg Body Composition & Open Label Extension Evaluate tolerability with 2.5 mg starting dose Core Focus Key Questions Supplementary Studies Is there weight loss plateau beyond 36 weeks? Can we dose higher than 120 mg? Will lower 2.5 mg starting dose further improve tolerability?
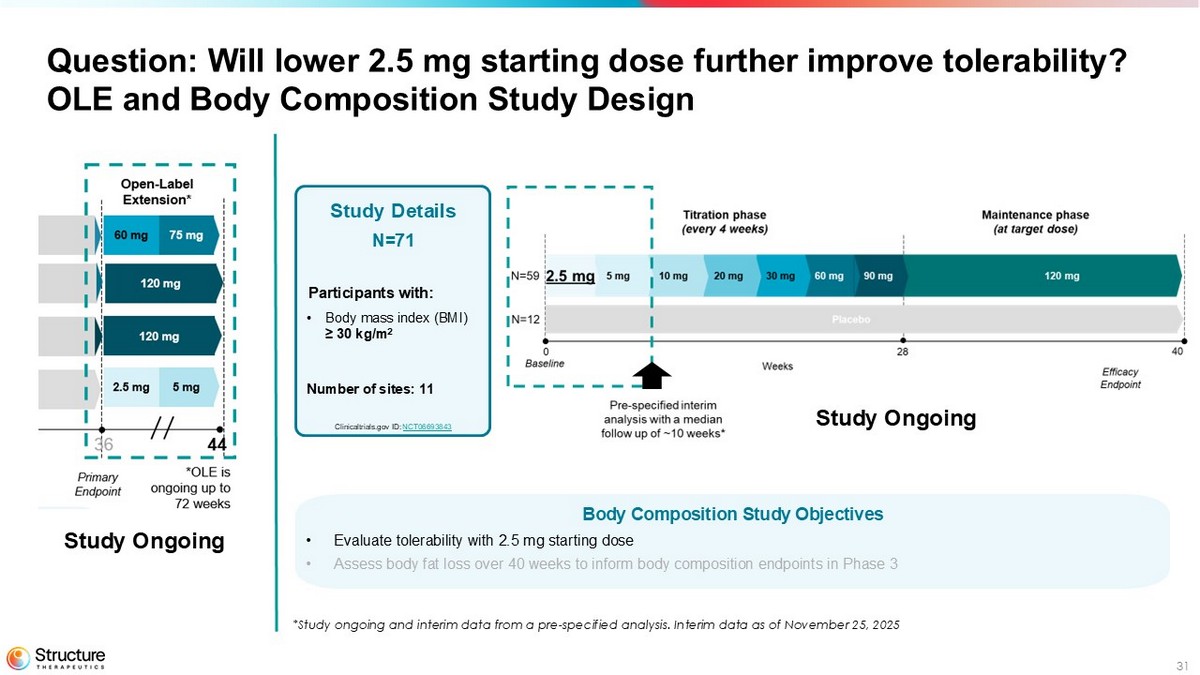
31 Question: Will lower 2.5 mg starting dose further improve tolerability? OLE and Body Composition Study Design Body Composition Study Objectives • Evaluate tolerability with 2.5 mg starting dose • Assess body fat loss over 40 weeks to inform body composition endpoints in Phase 3 Study Details N=71 Participants with: • Body mass index (BMI) ≥ 30 kg/m 2 Number of sites: 11 Clinicaltrials.gov ID: NCT06693843 Study Ongoing *Study ongoing and interim data from a pre - specified analysis. Interim data as of November 25, 2025 Study Ongoing
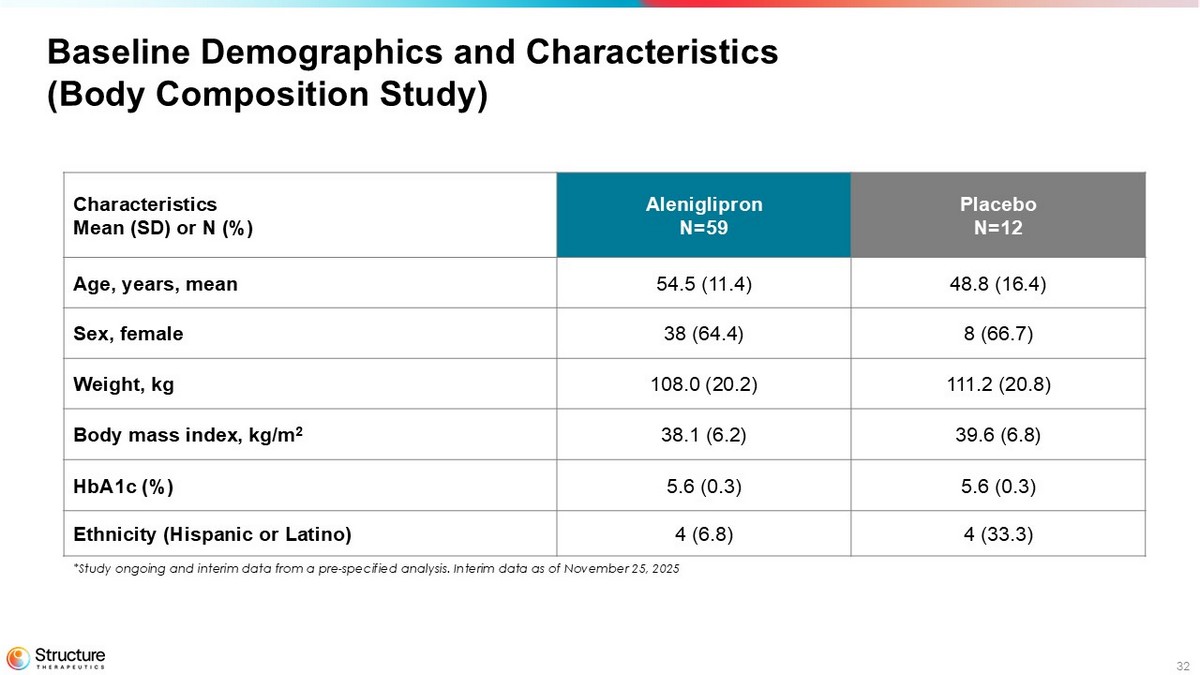
32 Baseline Demographics and Characteristics (Body Composition Study) Placebo N=12 Aleniglipron N=59 Characteristics Mean (SD) or N (%) 48.8 (16.4) 54.5 (11.4) Age, years, mean 8 (66.7) 38 (64.4) Sex, female 111.2 (20.8) 108.0 (20.2) Weight, kg 39.6 (6.8) 38.1 (6.2) Body mass index, kg/m 2 5.6 (0.3) 5.6 (0.3) HbA1c (%) 4 (33.3) 4 (6.8) Ethnicity (Hispanic or Latino) *Study ongoing and interim data from a pre - specified analysis. Interim data as of November 25, 2025
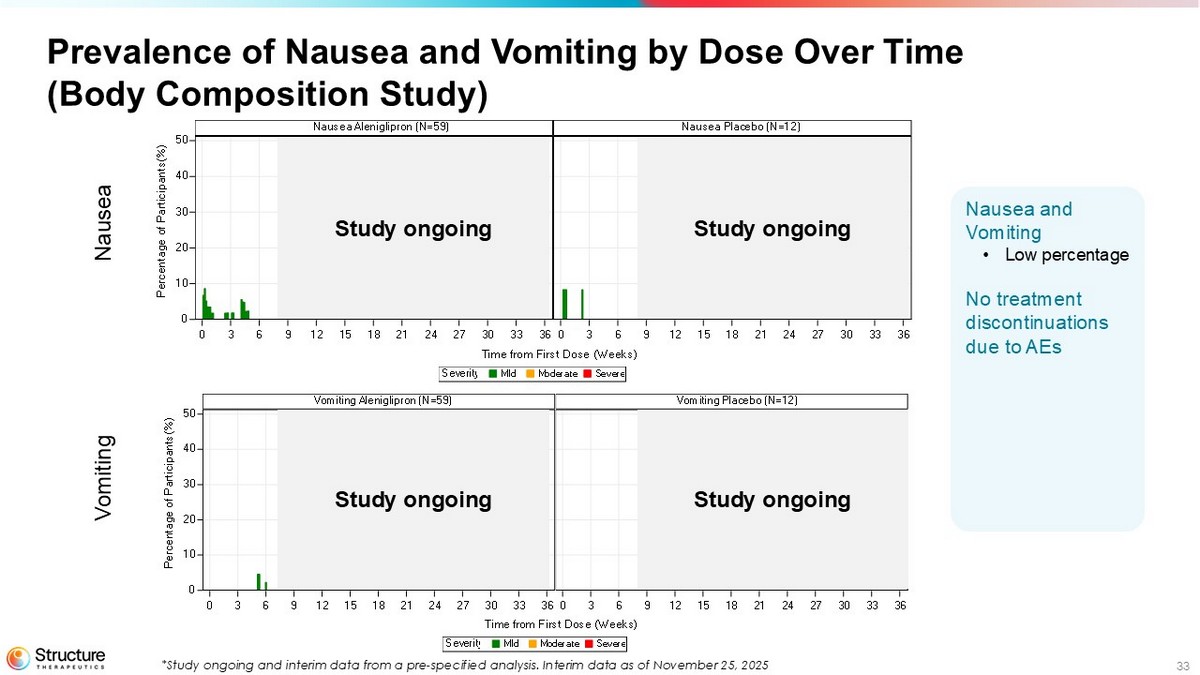
33 Prevalence of Nausea and Vomiting by Dose Over Time (Body Composition Study) SevereModerateMildSeverity Time from First Dose (Weeks) Nausea Placebo (N=12)Nausea Aleniglipron (N=59) 0 3 6 9 12 15 18 21 24 27 30 33 360 3 6 9 12 15 18 21 24 27 30 33 36 0 10 20 30 40 50 P e r c e n t a g e o f P a r t i c i p a n t s ( % ) SevereModerateMildSeverity Time from First Dose (Weeks) Vomiting Placebo (N=12)Vomiting Aleniglipron (N=59) 0 3 6 9 12 15 18 21 24 27 30 33 360 3 6 9 12 15 18 21 24 27 30 33 36 0 10 20 30 40 50 P e r c e n t a g e o f P a r t i c i p a n t s ( % ) Nausea Vomiting Study ongoing Study ongoing Study ongoing Study ongoing Nausea and Vomiting • Low percentage No treatment discontinuations due to AEs *Study ongoing and interim data from a pre - specified analysis. Interim data as of November 25, 2025

34 Cumulative Aleniglipron Tolerability Results with 2.5 mg Starting Dose (Body Composition Study) Study Ongoing Placebo N=12 Aleniglipron N=59 N (%) Reporting at least one event 2.5 mg Starting Dose 9 (75.0) 42 (71.2) Any TEAE 0 0 Any TEAE leading to discontinuation of treatment 2 (16.7) 21 (35.6) Nausea 0 5 (8.5) Vomiting 3 (25.0) 11 (18.6) Diarrhea 3 (25.0) 15 (25.4) Constipation • No treatment discontinuations due to AEs after a median treatment of ~10 weeks * *Study ongoing and interim data from a pre - specified analysis. Interim data as of November 25, 2025
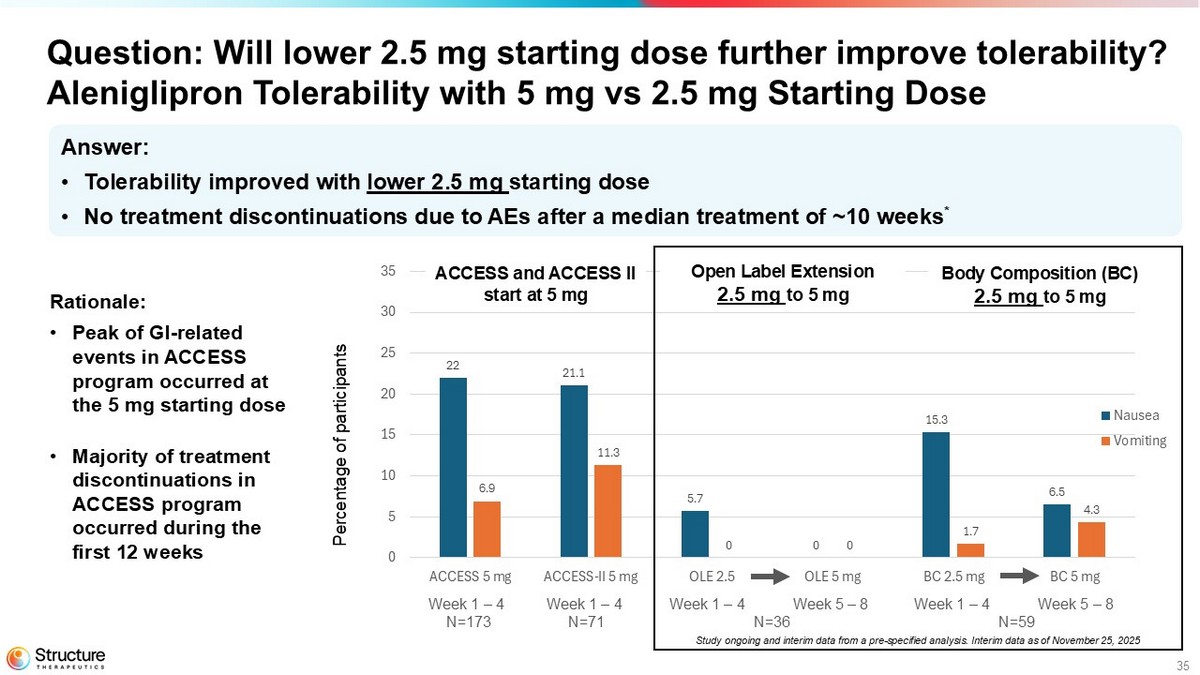
35 22 21.1 5.7 0 15.3 6.5 6.9 11.3 0 0 1.7 4.3 0 5 10 15 20 25 30 35 40 45 50 ACCESS 5 mg ACCESS-II 5 mg OLE 2.5 OLE 5 mg BC 2.5 mg BC 5 mg Nausea Vomiting Question: Will lower 2.5 mg starting dose further improve tolerability? Aleniglipron Tolerability with 5 mg vs 2.5 mg Starting Dose Percentage of participants Open Label Extension 2.5 mg to 5 mg ACCESS and ACCESS II start at 5 mg Body Composition (BC) 2.5 mg to 5 mg Week 1 – 4 N=173 Week 1 – 4 N=71 Week 1 – 4 Week 5 – 8 N=36 Week 1 – 4 Week 5 – 8 N=59 Answer: • Tolerability improved with lower 2.5 mg starting dose • No treatment discontinuations due to AEs after a median treatment of ~10 weeks * Study ongoing and interim data from a pre - specified analysis. Interim data as of November 25, 2025 Rationale: • Peak of GI - related events in ACCESS program occurred at the 5 mg starting dose • Majority of treatment discontinuations in ACCESS program occurred during the first 12 weeks

36 Aleniglipron Off - Target Safety Results Blai Coll
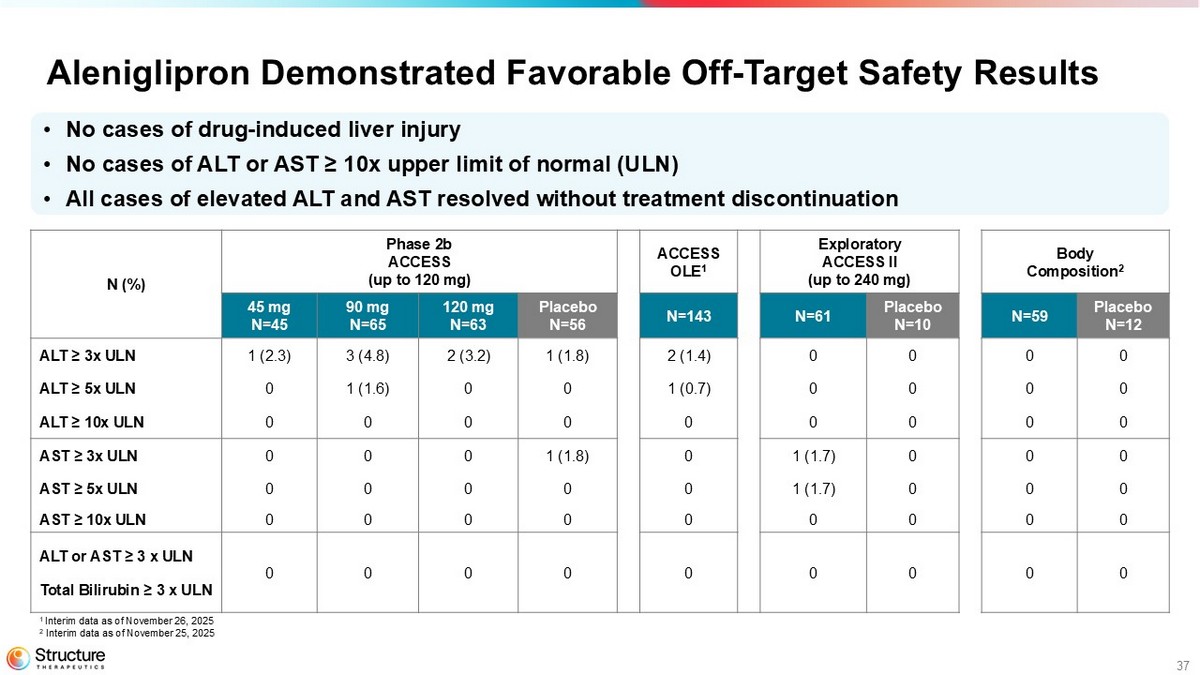
37 Aleniglipron Demonstrated Favorable Off - Target Safety Results Body Composition 2 Exploratory ACCESS II (up to 240 mg) ACCESS OLE 1 Phase 2b ACCESS (up to 120 mg) N (%) Placebo N=12 N=59 Placebo N=10 N=61 N=143 Placebo N=56 120 mg N=63 90 mg N=65 45 mg N=45 0 0 0 0 2 (1.4) 1 (1.8) 2 (3.2) 3 (4.8) 1 (2.3) ALT ≥ 3x ULN 0 0 0 0 1 (0.7) 0 0 1 (1.6) 0 ALT ≥ 5x ULN 0 0 0 0 0 0 0 0 0 ALT ≥ 10x ULN 0 0 0 1 (1.7) 0 1 (1.8) 0 0 0 AST ≥ 3x ULN 0 0 0 1 (1.7) 0 0 0 0 0 AST ≥ 5x ULN 0 0 0 0 0 0 0 0 0 AST ≥ 10x ULN 0 0 0 0 0 0 0 0 0 ALT or AST ≥ 3 x ULN Total Bilirubin ≥ 3 x ULN • No cases of drug - induced liver injury • No cases of ALT or AST ≥ 10x upper limit of normal (ULN) • All cases of elevated ALT and AST resolved without treatment discontinuation 1 Interim data as of November 26, 2025 2 Interim data as of November 25, 2025

38 Aleniglipron Obesity Data Summary Differentiated Selective Oral GLP - 1R Small Molecule Agonist Safety • > 500 participants treated across all studies up to 44 weeks o No events of drug induced liver injury o No off - target safety signals across all dose levels o No events of QTc prolongation Tolerability • Phase 2b ACCESS : o GI - related AEs consistent with GLP - 1RA class o Overall 10.4% AE - related treatment discontinuations • Exploratory ACCESS II : o For those participants who achieved re - randomization – No AE - related treatment discontinuations up to 240 mg dose • Open Label Extension (OLE) & Body Composition: o Clinically meaningful improvement in tolerability when starting at 2.5 mg compared to 5 mg start o No discontinuations observed to date* Efficacy • Phase 2b ACCESS 36 week placebo - adjusted mean weight loss: o 8.2% at 45 mg o 9.8% at 90 mg o 11.3% at 120 mg • Exploratory ACCESS II 36 week p lacebo - adjusted mean weight loss: o 14.1% at 120 mg o 14.4% at 180 mg o 15.3% at 240 mg • No evidence of weight loss plateau • Proportional pharmacokinetic (PK) exposure up to 240 mg • Clinically meaningful improvements in blood pressure and HbA1c *Study ongoing and interim data from a pre - specified analysis. Interim data as of November 25, 2025

39 Aleniglipron Phase 3 Readiness Blai Coll
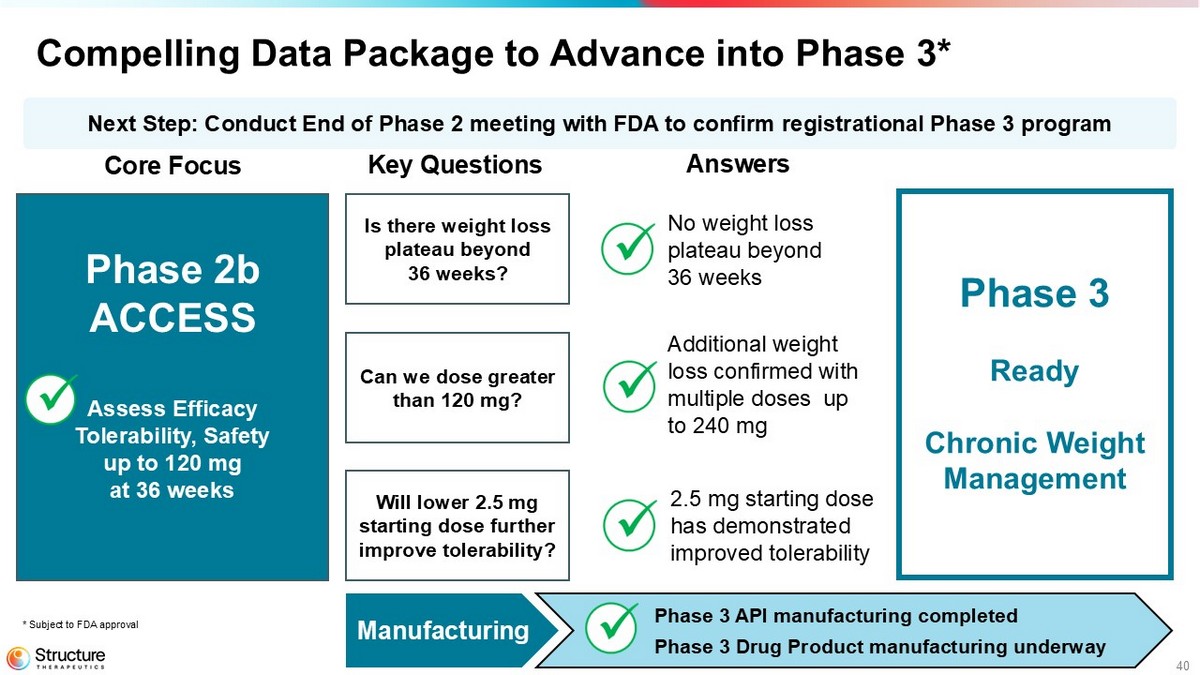
40 Compelling Data Package to Advance into Phase 3* Manufacturing Phase 3 API manufacturing completed Phase 3 Drug Product manufacturing underway Next Step: Conduct End of Phase 2 meeting with FDA to confirm registrational Phase 3 program Phase 2b ACCESS Assess Efficacy Tolerability, Safety up to 120 mg at 36 weeks Core Focus Key Questions Answers Is there weight loss plateau beyond 36 weeks? Can we dose greater than 120 mg? Will lower 2.5 mg starting dose further improve tolerability? Phase 3 Ready Chronic Weight Management * Subject to FDA approval 2.5 mg starting dose has demonstrated improved tolerability No weight loss plateau beyond 36 weeks Additional weight loss confirmed with multiple doses up to 240 mg

41 Aleniglipron: A Backbone in Structure's Oral Small Molecule Portfolio Raymond Stevens

42 Aleniglipron is Differentiated and is Well Positioned to Capture a Significant Proportion of the Growing Obesity Market 2025 2026 2027 2028 2029 2030 Specialists Non - specialists Non - specialists = 75% Rx today and growing 3 Only > 5M patients on incretin treatment today in US 2 1. World Obesity Atlas 2024 https://s3 - eu - west - 1.amazonaws.com/wof - files/WOF_Obesity_Atlas_2024.pdf 2. August 2025 Symphony Health prescription data and 2025 Evaluate Pharma sales data 3 Kumar et al., ISPOR (2025). https://www.ispor.org/docs/default - source/cti - meeting - 21021 - documents/6fe2eb97 - d003 - 4dc0 - b6d0 - 812 e13795055.pdf?sfvrsn=45a94bc8_0. Based on July 2025 TRx data for Wegovy. Non - specialists include PCPs, Nurse Practitioners, Phys ician Assistants, and Family Medicine physicians 4 Based on internal physician survey and interview data 5.Triangulation between internal data, analog disease areas, and 3rd party estimates; • PCPs/non - specialists indicate higher preference for orals due to convenience & potential for broader access and coverage 4 • Orals represent an exciting new option for long - term maintenance; more optionality for patients could facilitate improved persistence • Oral small molecules well - positioned to address unmet needs; 25 - 50% of market could be orals 5 • 15.3% placebo - adjusted mean weight loss at 36 weeks with 240 mg • Proportional exposure up to 240 mg • No evidence of weight loss plateau • 2.5 mg start further improves tolerability • Scaleable to meet global demand Only oral small molecules can scale and meet the needs of the global obesity patient population, estimated to be 1.5 billion by 2030 1 GLP - 1R Aleniglipron
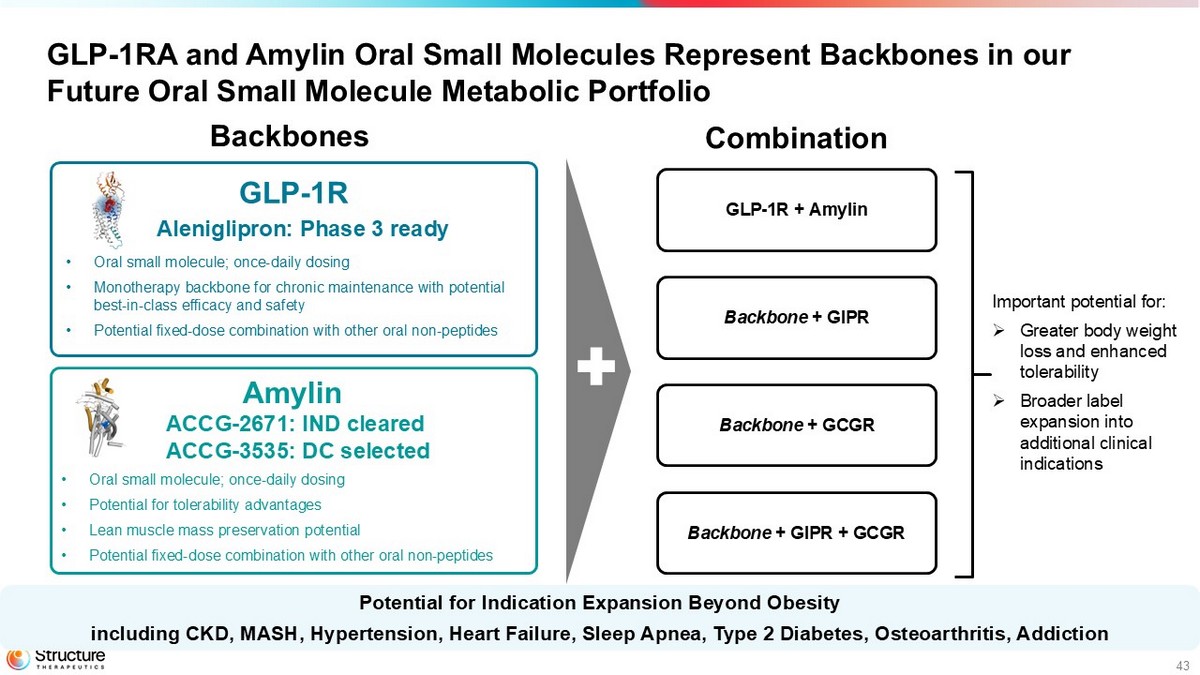
43 GLP - 1RA and Amylin Oral Small Molecules Represent Backbones in our Future Oral Small Molecule Metabolic Portfolio Backbone + GCGR GLP - 1R + Amylin Backbones Combination Backbone + GIPR + GCGR Backbone + GIPR • Oral small molecule; once - daily dosing • Monotherapy backbone for chronic maintenance with potential best - in - class efficacy and safety • Potential fixed - dose combination with other oral non - peptides GLP - 1R • Oral small molecule; once - daily dosing • Potential for tolerability advantages • Lean muscle mass preservation potential • Potential fixed - dose combination with other oral non - peptides Potential for Indication Expansion Beyond Obesity including CKD, MASH, Hypertension, Heart Failure, Sleep Apnea, Type 2 Diabetes, Osteoarthritis, Addiction Aleniglipron: Phase 3 ready Amylin ACCG - 2671: IND cleared ACCG - 3535 : DC selected Important potential for: » G reater body weight loss and enhanced tolerability » B roader label expansion into additional clinical indications
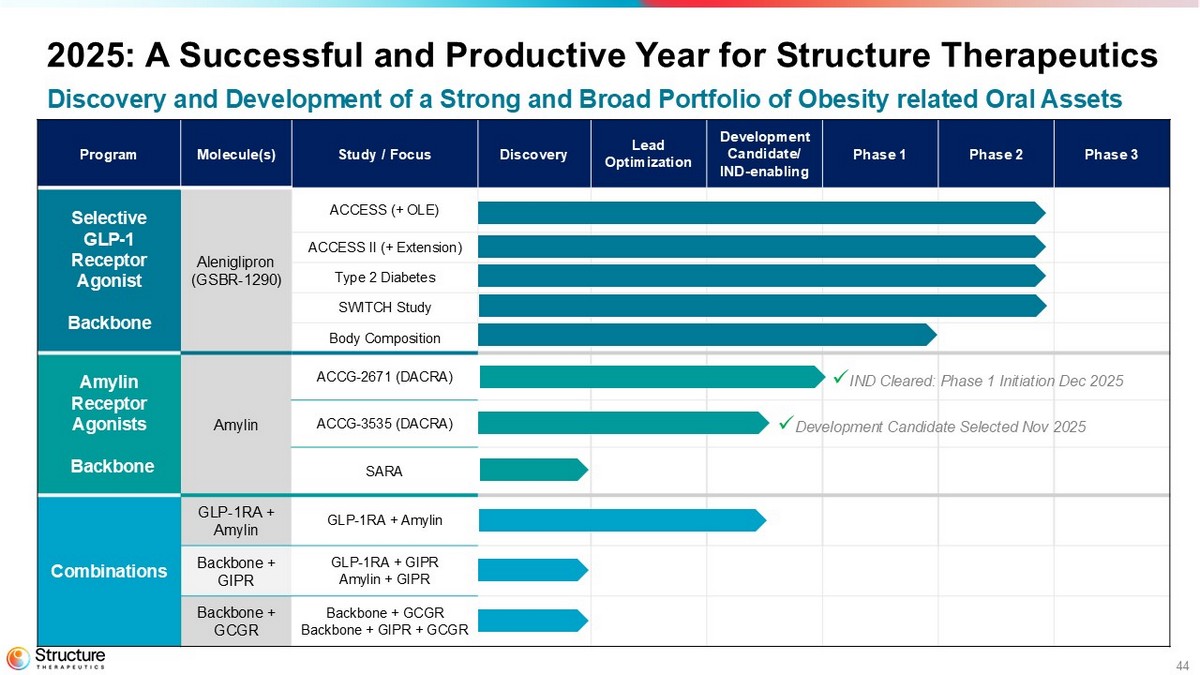
44 Phase 3 Phase 2 Phase 1 Development Candidate/ IND - enabling Lead Optimization Discovery Study / Focus Molecule(s) Program ACCESS (+ OLE) Aleniglipron (GSBR - 1290) Selective GLP - 1 Receptor Agonist Backbone ACCESS II (+ Extension) Type 2 Diabetes SWITCH Study Body Composition ACCG - 2671 (DACRA) Amylin Amylin Receptor Agonists Backbone ACCG - 3535 (DACRA) SARA GLP - 1RA + Amylin GLP - 1RA + Amylin Combinations GLP - 1RA + GIPR Amylin + GIPR Backbone + GIPR Backbone + GCGR Backbone + GIPR + GCGR Backbone + GCGR x IND Cleared: Phase 1 I nitiation Dec 2025 x Development Candidate Selected Nov 2025 2025: A Successful and Productive Year for Structure Therapeutics Discovery and Development of a Strong and Broad Portfolio of Obesity related Oral Assets
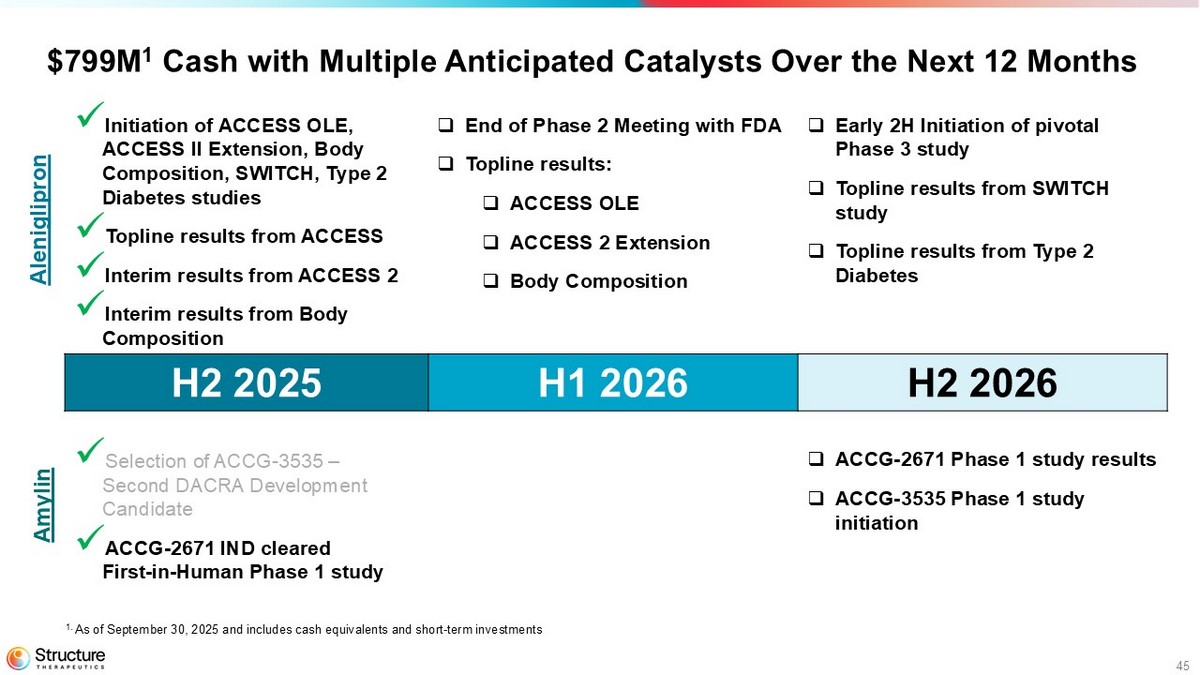
45 $799M 1 Cash with Multiple Anticipated Catalysts Over the Next 12 Months □ Early 2H Initiation of pivotal Phase 3 study □ Topline results from SWITCH study □ Topline results from Type 2 Diabetes □ End of Phase 2 Meeting with FDA □ Topline results: □ ACCESS OLE □ ACCESS 2 Extension □ Body Composition x Initiation of ACCESS OLE, ACCESS II Extension, Body Composition, SWITCH, Type 2 Diabetes studies x Topline results from ACCESS x Interim results from ACCESS 2 x Interim results from Body Composition H2 2026 H1 2026 H2 2025 □ ACCG - 2671 Phase 1 study results □ ACCG - 3535 Phase 1 study initiation x Selection of ACCG - 3535 – Second DACRA Development Candidate x ACCG - 2671 IND cleared First - in - Human Phase 1 study Aleniglipron Amylin 1. As of September 30, 2025 and includes cash equivalents and short - term investments

46 Our Mission Making medicines more accessible to all

47 Q&A
